UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 8-K
CURRENT REPORT
Pursuant to Section 13 or 15(d)
of the Securities Exchange Act of 1934
Date of Report (Date of earliest event reported): December 8, 2025
(Exact name of registrant as specified in its charter)
| (State or other jurisdiction of incorporation) | (Commission File Number) | (I.R.S. Employer Identification No.) | ||||||||||||
(Address of principal executive offices) (Zip code)
(385 ) 269 - 0203
(Registrant’s telephone number, including area code)
Not Applicable
(Former name or former address, if changed since last report.)
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions:
Securities registered pursuant to Section 12(b) of the Act:
| Title of each class | Trading symbol(s) | Name of each exchange on which registered | ||||||
Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 or (§230.405 of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§240.12b-2 of this chapter).
Emerging growth company ☐
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Item 8.01. Other Events.
On December 8, 2025, Recursion Pharmaceuticals, Inc. (“Recursion” or the “Company”) issued a press release announcing Phase 1b/2 data from the ongoing TUPELO trial of REC-4881, an investigational allosteric MEK1/2 inhibitor for familial adenomatous polyposis (FAP). A copy of the press release is attached hereto as Exhibit 99.1 and incorporated by reference herein.
Also on December 8, 2025, the Company released a presentation of the data from the TUPELO Trial. A copy of the presentation is attached hereto as Exhibit 99.2 and incorporated herein by reference.
Item 9.01. Financial Statements and Exhibits.
(d) Exhibits
| Exhibit Number | Description | ||||
| 99.1 | |||||
| 99.2 | |||||
| 104 | Cover Page Interactive Data File (embedded within the Inline XBRL document) | ||||
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized on December 8, 2025.
| RECURSION PHARMACEUTICALS, INC. | ||||||||
| By: | /s/ Nathan Hatfield | |||||||
Nathan Hatfield | ||||||||
| Chief Legal Officer | ||||||||
Exhibit 99.1
Positive Phase 1b/2 Results from Ongoing REC-4881 TUPELO Trial Demonstrate Rapid and Durable Reductions in Polyp Burden in Familial Adenomatous Polyposis (FAP) at 25 Weeks
●REC-4881 (4 mg QD) achieved rapid clinical activity, with 75% of evaluable patients showing reductions in total polyp burden and a 43% median reduction after 12 weeks of treatment (n=12)
●After 12 weeks off therapy (week 25 of the study), 82% of evaluable patients (9 of 11) maintained a durable reduction in total polyp burden, with a 53% median reduction observed from baseline
●Natural history analysis showed that 87% of untreated FAP patients - who resembled the inclusion criteria of TUPELO - had annualized polyp-burden increase, 10% remained stable, and 3% showed modest decrease—underscoring the disease’s progressive trajectory (n=55)
●40% of patients (4 out of 10) achieved a ≥1-point improvement in Spigelman stage—a clinically meaningful measure of upper GI disease severity to assess surveillance and clinical management
●REC-4881 (4 mg QD) has a safety profile consistent with MEK1/2 inhibition, with the majority of treatment-related adverse events being Grade 1 or 2, Grade 3 events occurring in 15.8% of the safety-evaluable patients, and no Grade ≥4 TRAEs reported to date
●First clinical validation of the Recursion OS, demonstrating how unbiased phenotypic and mechanistic insights—such as MEK1/2 rescue of APC loss-of-function—can translate to novel, differentiated therapeutics for diseases like FAP with no approved therapy and high prevalence of 50,000 patients in US and EU5
●Next steps: Engage the FDA in the 1H26 to define a potential registration pathway, and in parallel, expand the population from ≥55 to ≥18 years old, and further optimize dosing schedule
SALT LAKE CITY, December 8, 2025 (GLOBE NEWSWIRE) — Recursion (Nasdaq: RXRX), a clinical-stage TechBio company decoding biology to radically improve lives, today announced positive Phase 1b/2 data from the ongoing TUPELO trial of REC-4881, an investigational allosteric MEK1/2 inhibitor for familial adenomatous polyposis (FAP).
Through an unbiased phenotypic screen of thousands of compounds, the earliest version of the Recursion OS identified selective MEK1/2 inhibition as a highly specific mechanism capable of reversing APC loss-of-function signatures. Using high-content cellular phenomics driven by AI, REC-4881 emerged as one of the strongest phenotypic rescue hits, reverting APC-deficient cells toward a healthy state and suppressing ERK/MAPK hyperactivation downstream of APC loss. Guided by this AI-driven insight, Recursion in-licensed REC-4881 from Takeda and redirected REC-4881—originally evaluated clinically in solid tumors—as a mechanistically aligned therapeutic candidate for FAP. REC-4881 is now the first MEK1/2 inhibitor ever studied clinically for this disease.
“The durable polyp-burden reduction demonstrated by REC-4881—especially the sustained effect seen at Week 25, 12 weeks after completing therapy—is highly encouraging for the FAP community,” said Jessica Stout, D.O., Assistant Clinical Professor, University of Utah School of Medicine, and Principal Investigator of the TUPELO study. “Given the near-100% lifetime risk of colorectal cancer and the absence of any approved medical therapies, patients today often face a lifetime of intensive surveillance and life-altering surgeries. These Phase 2 results provide a meaningful basis for hope and support the potential for REC-4881 to offer a much-needed non-surgical option for this debilitating, life-long disease.”
“These Phase 2 results mark a meaningful validation of the Recursion OS,” said Chris Gibson, Ph.D., Co-Founder and CEO of Recursion. “An unbiased phenotypic insight from our platform and driven by AI—linking MEK1/2 inhibition to APC loss-of-function biology—has now translated into rapid, substantial, and
durable reductions in polyp burden in patients. This is a powerful example of how even the earliest versions of the Recursion OS can uncover therapeutic opportunities in diseases with no approved pharmacotherapy options. And since this discovery, we’ve only added to the breadth, depth, and power of the Recursion OS; we believe this is the first of many potential medicines that will advance as our flywheel of discovery accelerates”.
In the Phase 2 portion of the study, REC-4881 demonstrated rapid and durable reductions in polyp burden, with 43% median reduction in evaluable patients after 12 weeks of treatment. 75% of evaluable patients had a reduction in polyp burden in this same period. Importantly, the effect persisted well beyond the dosing period: 82% of evaluable patients (9 of 11) maintained reductions at Week 25—12 weeks after stopping therapy—with a 53% median decrease from baseline. These results are especially meaningful when considered alongside real-world natural history analyses showing that untreated FAP patients are expected to experience increases when left untreated .
“This program reflects a full validation cycle of the Recursion OS: an unbiased phenotypic signal identifying MEK1/2 inhibition as a rescue mechanism for APC loss-of-function, followed by mechanistic confirmation, clinical translation, and now encouraging human data in a disease with no approved medical therapies,” said Najat Khan, Ph.D., Chief R&D and Commercial Officer and incoming President and CEO. “REC-4881, an allosteric MEK1/2 inhibitor, represents a first precision-medicine approach for the causal biology of FAP. In TUPELO, we are seeing rapid, substantial, and durable reductions in polyp burden — including sustained benefit after patients stop therapy. Equally important, our ClinTech and real-world data capabilities have been instrumental in guiding this program — from refining eligibility to contextualizing a single-arm dataset with a first-of-its-kind natural history study”.
About FAP
FAP is one of the most clinically significant hereditary colorectal cancer syndromes and is caused by inactivating mutations in the APC gene, leading to the growth of hundreds to thousands of gastrointestinal polyps and a near 100% risk of developing colorectal cancer before the age of 40 if left untreated. With no approved pharmacotherapies, excisions followed sequentially by debilitating, life altering surgeries remain the only option to remove polyps and polyp burdened organs, typically involving colectomy in the early 20s. While this procedure removes immediate colon cancer risk, it does not address the underlying disease biology and does not prevent future polyp formation. Patients will continue to develop polyps in the rectum or upper GI tract, with approximately 50% of patients requiring subsequent life-altering surgical procedures to manage the persistent disease. Current treatment approaches lead to substantial long-term loss of quality of life, affecting continence, fertility, and long-term gastrointestinal function in relatively young patients. Approximately 90% of FAP patients go on to develop duodenal adenomas, with 6% eventually undergoing a high-morbidity risk duodenectomy to control polyp growth. FAP affects an estimated 50,000 individuals across the US and EU5 (France, Germany, Italy, Spain, and the UK).
Background on REC-4881 and Recursion’s Platform Insights
REC-4881 was discovered using one of the earliest versions of the Recursion OS (v0.1), through an unbiased, high-content AI-driven phenotypic screening approach in APC-deficient human cell models. Because FAP is driven by loss-of-function mutations in the APC gene, the platform was designed to identify molecules capable of rescuing APC-dependent biology—guided entirely by cellular phenotype, without presupposing any specific mechanism. Using AI/ML to extract and compare over a thousand morphological features that distinguish “diseased” from “healthy” states, the Recursion OS screened numerous investigative compounds and identified REC-4881 as one of the most robust phenotype-rescuing hits. In follow-up assays, REC-4881 consistently reverted APC-deficient cells toward a healthy-state phenotype and demonstrated potent, selective, and concentration-dependent MEK1/2 inhibition that was not seen across hundreds of other oncogenes and tumor suppressor models tested.
Importantly, the Recursion OS highlighted MEK1/2 inhibition as a mechanistic strategy to exploit a therapeutic vulnerability arising from APC loss in FAP—a disease area where MEK1/2 inhibition had not previously been investigated as a therapeutic strategy in a clinical setting. Based on this novel insight, Recursion in-licensed REC-4881 from Takeda, where it had been evaluated in solid tumors, and redirected it as the first MEK1/2 inhibitor advanced clinically for FAP. This program represents the power of a phenotype-first AI-driven discovery model: the platform surfaced a mechanistically aligned therapeutic opportunity solely through scaled high-dimensional exploration.
Today, the Company is using the Recursion OS 2.0 platform—including proprietary ClinTech capabilities of large-scale real-world evidence (RWE) analytics—to further advance the REC-4881 program. This includes a comprehensive natural history collaboration with Amsterdam University Medical Center, home to one of the largest and longest-running FAP registries, as well as analysis of more than 1,000 US FAP patients and 250,000 physician notes processed through Recursion’s custom LLM-based pipeline. Together, these data reinforce the relentlessly progressive nature of FAP, highlight the absence of spontaneous polyp regression, and demonstrate the substantial burden of repeated polyp-removal procedures and major surgeries experienced by real-world patients.
ClinTech insights also helped refine the design of the ongoing TUPELO trial, including expanding age eligibility from ≥55 to ≥18. This expansion was based on a thorough risk-benefit analysis, enabling evaluation of REC-4881 in younger patients who represent a substantial portion of FAP patients. REC-4881 has received Fast Track and Orphan Drug designations from the US FDA, as well as Orphan Drug designation from the European Commission.
Results of the Phase 2 Data for Ongoing REC-4881 Trial
Efficacy and Durability Findings
As of the November 25, 2025 data cutoff in the open-label Phase 2 portion of TUPELO, treatment with REC-4881 (4 mg QD) demonstrated meaningful and durable reductions in polyp burden in patients with FAP.
Week 13 Assessment
●REC-4881 produced a median 43% reduction in total polyp burden among 12 efficacy-evaluable patients.
●The majority of evaluable patients responded, with 75% showing reductions in polyp burden after 12 weeks of therapy.
●40% of patients (4 out of 10) achieved a ≥1-point improvement in Spigelman stage—a clinically meaningful measure of upper GI disease severity to assess surveillance and clinical management.
●Other investigational agents currently under evaluation in separate studies generated approximately 17–29% median reduction in polyp burden after 12 months of treatment; no off-treatment durability was reported (Biodexa press release, June 24 2024)
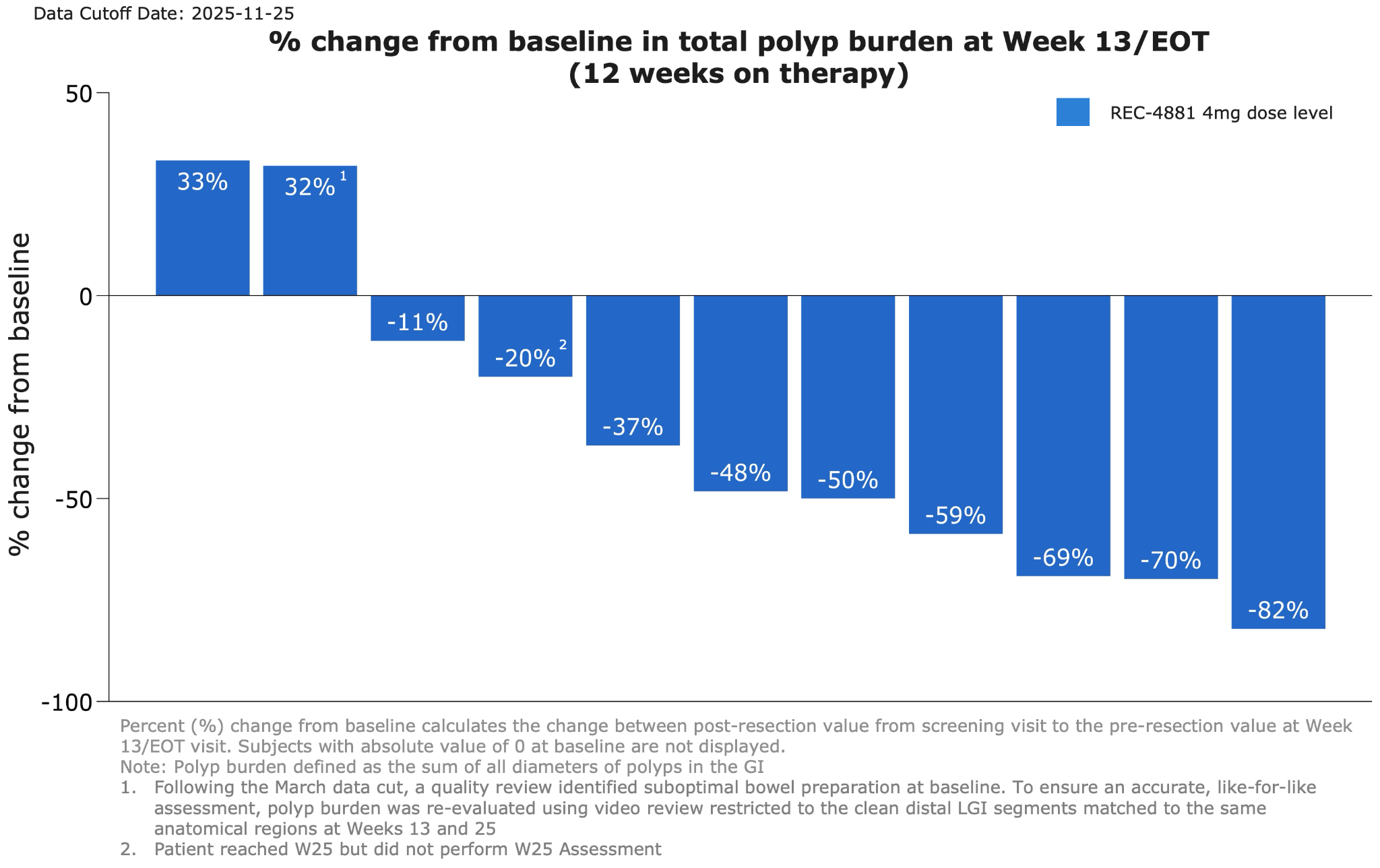
Figure 1: Waterfall plot showing percent change from baseline in total polyp burden at Week 13 for efficacy-evaluable patients receiving REC-4881 (4 mg QD)
Week 25 Assessment
●After 12 weeks of treatment, patients went off treatment for an additional 12-weeks. Durability of effect was maintained during the off-treatment observation period with 82% of patients responding (>0% reduction; 9 out of 11) at Week 25.
●73% achieved durable ≥30% reductions in polyp burden with a 53% median reduction in total polyp burden observed.
●40% of patients (4 out of 10) maintained a ≥1-point improvement in Spigelman stage from baseline.
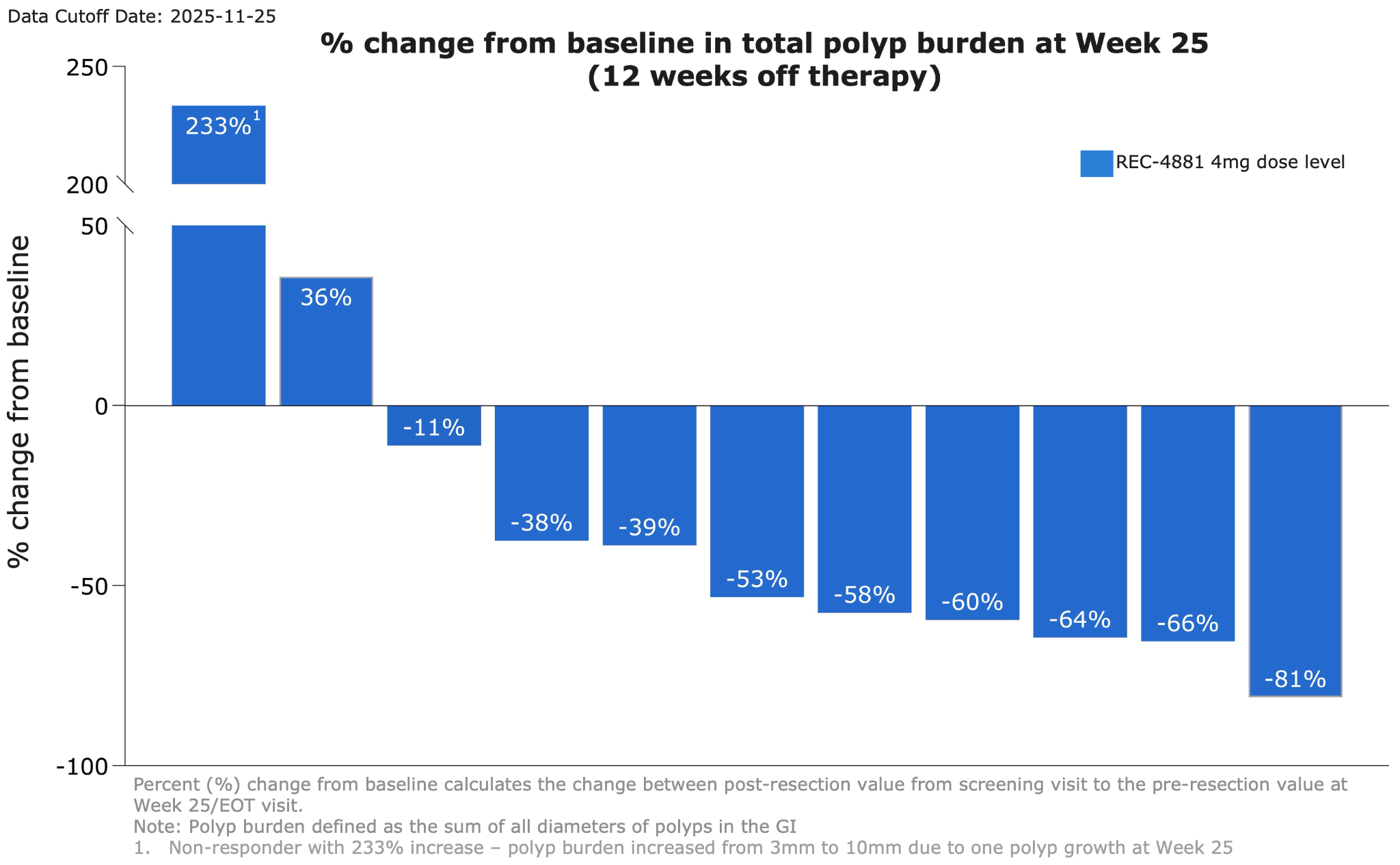
Figure 2: Waterfall plot showing percent change from baseline in total polyp burden at Week 25 for efficacy-evaluable patients receiving REC-4881 (4 mg QD)
Safety Summary
As of the data cutoff, treatment with 4 mg REC-4881 demonstrated a safety profile generally consistent with prior MEK1/2 inhibitors.
●Across the combined Phase 1b and Phase 2 safety cohorts (n=19), 94.7% of patients reported at least one treatment-related adverse event (TRAE), the majority of which were Grade 1/2 in severity. The most frequent TRAEs (≥10%) included: dermatitis acneiform / rash and blood CPK increase.
●Grade 3 TRAEs occurred in 15.8% of patients; no Grade ≥4 TRAEs have been reported to date.
●Treatment modifications were infrequent, with 2 of 19 patients experiencing dose interruption.
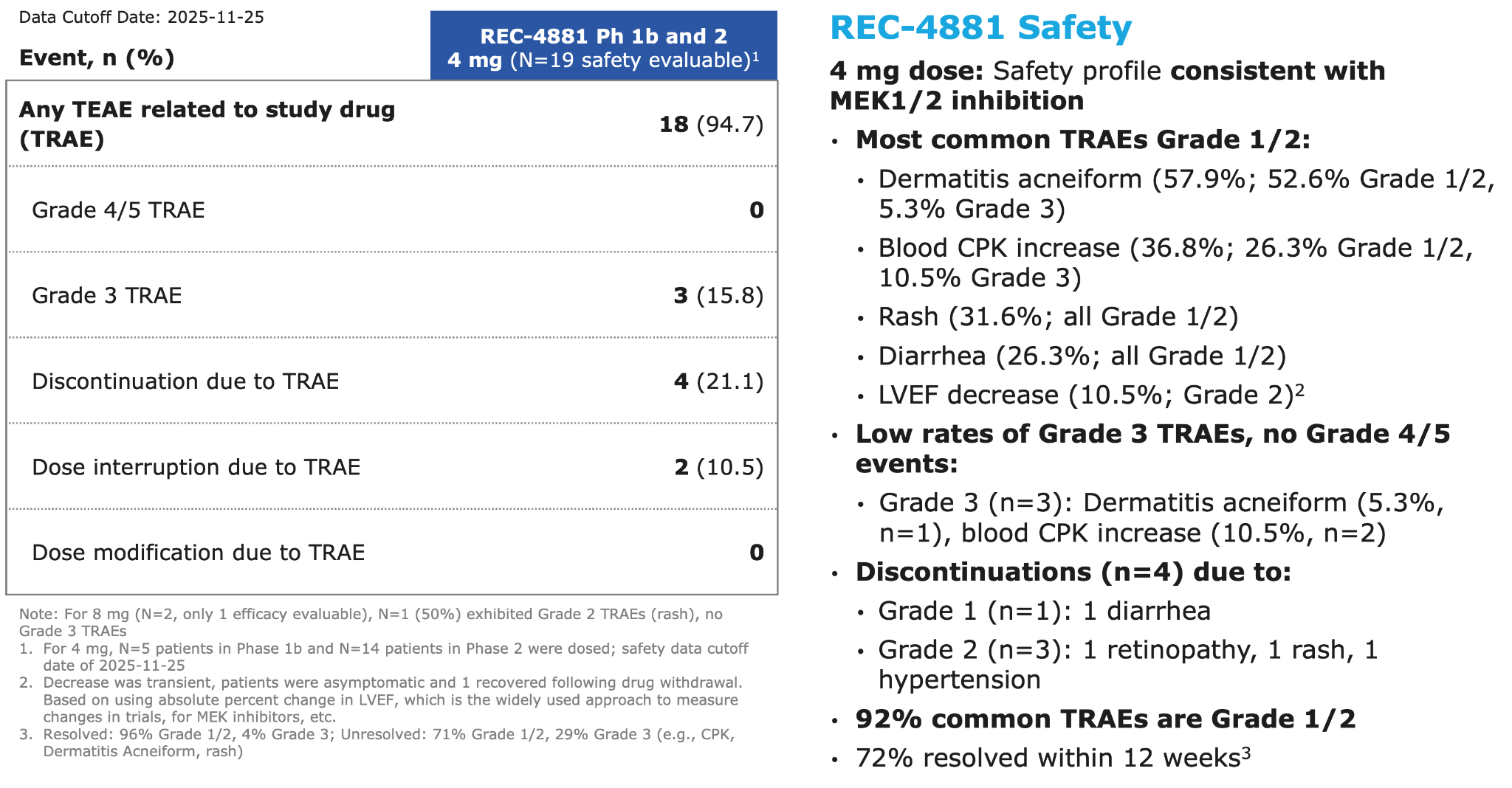
Figure 3: Summary of adverse events across Phase 1b and Phase 2 of the TUPELO trial
About the TUPELO Trial Design and Expanded Population
The Phase 1b/2 TUPELO trial is evaluating the safety, tolerability, pharmacokinetics (PK), and efficacy of REC-4881 monotherapy in patients with familial adenomatous polyposis (FAP).
Study Design and Analysis
Efficacy is assessed via upper and lower endoscopy at baseline, Week 13 (on-treatment), and Week 25 (off-treatment).
●The primary endpoint is percent change from baseline in polyp burden, which is the sum of all polyp diameters in the GI.
●The Efficacy Evaluable Population includes patients with measurable disease at baseline who received ≥75% of study drug and had at least one post-baseline endoscopic assessment.
Disease staging uses the Spigelman system for upper GI polyposis and the InSiGHT classification for the lower GI tract.
Disease staging uses the Spigelman system for upper GI polyposis and the InSiGHT classification for the lower GI tract.
Natural History Analyses
To better understand the natural history of FAP and to contextualize the single-arm efficacy of REC-4881, we collaborated with Amsterdam University Medical Center to analyze a registry of ~200 patients with FAP. 55 of these patients met the key inclusion criteria of TUPELO. We also leveraged our clinical development technology (ClinTech) platform to analyze US electronic health records (EHR), including physician notes, of ~1,000 FAP patients to assess disease progression and treatment patterns in the US. Both studies revealed high patient burden and progressive natural history of the disease. Results of the studies suggest that the natural history of FAP is to progress with relentless precancerous polyp progression: 87% of untreated patients in the registry experienced annualized increase in polyp burden. 10% were stable and 3% experienced a modest decrease in polyp burden. Mean increase of 60% and median increase of 28% in annualized polyp burden was observed. At least 75% of patients in the US EHR database had a major invasive surgery along with frequent polypectomies during follow-up.
Next Steps
Recursion plans to expand the population from ≥55 to ≥18 years old and further optimize dosing schedule. In parallel, the Company intends to engage the FDA in 1H26 to define a potential registration pathway.
Forward Looking Statements
This document contains information that includes or is based upon “forward-looking statements” within the meaning of the Securities Litigation Reform Act of 1995, including, without limitation, those regarding Recursion’s anticipated engagement with the FDA; the clinical relevance of the TUPELO trial data and obtaining additional confirmatory data; advancing potential transformational therapies for FAP and beyond; subsequent REC-4881 studies, including expanded enrollment and alternate dosing schedule, and their results and advancing Recursion’s REC-4881 program further; the size of the potential FAP patient population; Recursion OS and other technologies potential and advancement of the future of medicine; business and financial plans and performance; and all other statements that are not historical facts. Forward-looking statements may or may not include identifying words such as “plan,” “will,” “expect,” “anticipate,” “intend,” “believe,” “potential,” “continue,” and similar terms. These statements are subject to known or unknown risks and uncertainties that could cause actual results to differ materially from those expressed or implied in such statements, including but not limited to: challenges inherent in pharmaceutical research and development, including the timing and results of preclinical and clinical programs, where the risk of failure is high and failure can occur at any stage prior to or after regulatory approval due to lack of sufficient efficacy, safety considerations, or other factors; our ability to leverage and enhance our drug discovery platform; our ability to obtain financing for development activities and other corporate purposes; the success of our collaboration activities; our ability to obtain regulatory approval of, and ultimately commercialize, drug candidates; our ability to obtain, maintain, and enforce intellectual property protections; cyberattacks or other disruptions to our technology systems; our ability to attract, motivate, and retain key employees and manage our growth; inflation and other macroeconomic issues; and other risks and uncertainties such as those described under the heading “Risk Factors” in our filings with the U.S. Securities and Exchange Commission, including our Annual Report on Form 10-K and Quarterly Reports on Form 10-Q. All forward-looking statements are based on management’s current estimates, projections, and assumptions, and Recursion undertakes no obligation to correct or update any such statements, whether as a result of new information, future developments, or otherwise, except to the extent required by applicable law.
About Recursion
Recursion (NASDAQ: RXRX) is a clinical stage TechBio company leading the space by decoding biology to radically improve lives. Enabling its mission is the Recursion OS, a platform built across diverse technologies that continuously generate one of the world’s largest proprietary biological and chemical datasets. Recursion leverages sophisticated machine-learning algorithms to distill from its dataset a collection of trillions of searchable relationships across biology and chemistry unconstrained by human bias. By commanding massive experimental scale — up to millions of wet lab experiments weekly — and massive computational scale — owning and operating one of the most powerful supercomputers in the world, Recursion is uniting technology, biology and chemistry to advance the future of medicine.
Recursion (NASDAQ: RXRX) is a clinical stage TechBio company leading the space by decoding biology to radically improve lives. Enabling its mission is the Recursion OS, a platform built across diverse technologies that continuously generate one of the world’s largest proprietary biological and chemical datasets. Recursion leverages sophisticated machine-learning algorithms to distill from its dataset a collection of trillions of searchable relationships across biology and chemistry unconstrained by human bias. By commanding massive experimental scale — up to millions of wet lab experiments weekly — and massive computational scale — owning and operating one of the most powerful supercomputers in the world, Recursion is uniting technology, biology and chemistry to advance the future of medicine.
Recursion is headquartered in Salt Lake City, where it is a founding member of BioHive, the Utah life sciences industry collective. Recursion also has offices in Montréal, New York, London, and the Oxford area. Learn more at www.recursion.com, or connect on X and LinkedIn.
Media Contact
media@recursion.com
media@recursion.com
Investor Contact
investor@recursion.com
investor@recursion.com

Ongoing Phase 1b/2 Trial of the Allosteric MEK1/2 Inhibitor REC-4881 as Monotherapy in Familial Adenomatous Polyposis (FAP): Updated Safety and Efficacy December 8, 2025

This presentation discusses an investigational product whose safety and efficacy have not been established. This document contains information that includes or is based upon “forward-looking statements” within the meaning of the Securities Litigation Reform Act of 1995, including, without limitation, those regarding Recursion’s anticipated engagement w ith the FDA; the clinical relevance of the TUPELO trial data and obtaining additional confirmatory data; advancing potential transformational therapies for FAP and beyond; subsequent REC-4881 studies, including expanded enrollment and alternate dosing schedule, and their results and advancing Recursion’s REC-4881 program further; the size of the potential FAP patient population; Recursion OS and other technologies potential and advancement of the future of medicine; business and financial plans and performance; and all other statements that are not historical facts. Forward-looking statements may or may not include identifying words such as “plan,” “will,” “expect,” “anticipate,” “intend,” “believe,” “potential,” “continue,” and similar terms. These statements are subject to known or unknown risks and uncertainties that could cause actual results to differ materially from those expressed or implied in such statements, including but not limited to: challenges inherent in pharmaceutical research and development, including the timing and results of preclinical and clinical programs, where the risk of failure is high and failure can occur at any stage prior to or after regulatory approval due to lack of sufficient efficacy, safety considerations, or other factors; our ability to leverage and enhance our drug discovery platform; our ability to obtain financing for development activities and other corporate purposes; the success of our collaboration activities; our ability to obtain regulatory approval of, and ultimately commercialize, drug candidates; our ability to obtain, maintain, and enforce intellectual property protections; cyberattacks or other disruptions to our technology systems; our ability to attract, motivate, and retain key employees and manage our growth; inflation and other macroeconomic issues; and other risks and uncertainties such as those described under the heading “Risk Factors” in our filings with the U.S. Securities and Exchange Commission, including our Annual Report on Form 10-K and Quarterly Reports on Form 10-Q. All forward-looking statements are based on management’s current estimates, projections, and assumptions, and Recursion undertakes no obligation to correct or update any such statements, whether as a result of new information, future developments, or otherwise, except to the extent required by applicable law. Forward Looking Statements

From platform discovery to positive clinical data: REC-4881’s emerging potential for treatment of familial adenomatous polyposis High Unmet Need in FAP • >50,000 patients in US/EU5 • Lifelong surveillance and life altering surgeries • No approved pharmacotherapy Safety Data Update • Most common TRAEs in line with MEK1/2 class effect (incl. dermatitis acneiform, CPK increase, rash) • Most Grade 1/2, low rates of Grade 3, no grade 4/5 First Platform to Clinical Proof of Concept • Recursion’s unbiased AI-driven phenotypic approach uncovered MEK1/2i as a mechanism that rescues APC-deficient cells • First to clinically investigate MEK1/2 inhibition for FAP Rapid Polyp Burden Reduction • Meaningful polyp reduction at 4 mg in majority of patients after 12 weeks of treatment Durable Polyp Burden Reduction • Significant polyp burden reduction maintained in most patients after 12 weeks off- treatment Path Forward • Engage FDA to define a potential registration path (1H 2026) • Expand population from ≥55 to 18 years old and further optimize dosing schedule

4 Recursion momentum: Delivering pipeline advancements and partnership value in total cash inflows achieved across all our partnerships and collaborations >$500 million ✓ 6 Phenomaps: 4 GI oncology, 2 neuroscience ✓ 1 program initiated in GI onc indication ✓ 4 milestones achieved in 18 months ✓ Portfolio of projects continuing to expand ✓ Potential upcoming milestones (e.g., development candidate, lead series)
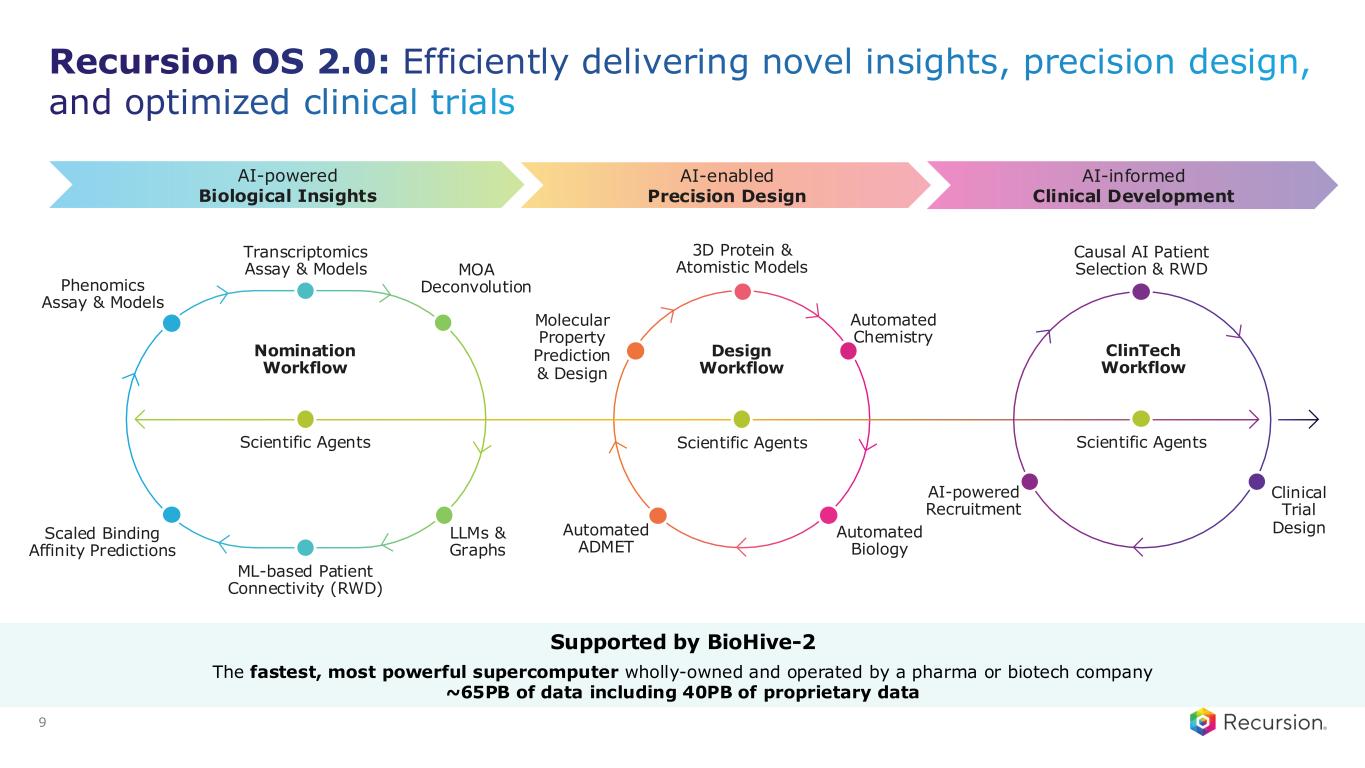
Design Workflow Automated ADMET Automated Chemistry Automated Biology 3D Protein & Atomistic Models Molecular Property Prediction & Design Scientific Agents Nomination Workflow Scientific Agents Transcriptomics Assay & Models ML-based Patient Connectivity (RWD) LLMs & Graphs MOA Deconvolution Scaled Binding Affinity Predictions Phenomics Assay & Models Clinical Trial Design ClinTech Workflow AI-powered Recruitment Causal AI Patient Selection & RWD AI-enabled Precision Design AI-powered Biological Insights AI-informed Clinical Development Scientific Agents Recursion OS 2.0: Efficiently delivering novel insights, precision design, and optimized clinical trials 9 Supported by BioHive-2 The fastest, most powerful supercomputer wholly-owned and operated by a pharma or biotech company ~65PB of data including 40PB of proprietary data

Causal AI Patient Selection & RWD Clinical Trial Design AI-powered Recruitment REC-4881: In-licensed based on novel insights from Recursion OS V0.1; now leveraging V2.0 platform to advance clinical development 9 Design Workflow Automated ADMET Automated Chemistry Automated Biology 3D Protein & Atomistic Models Molecular Property Prediction & Design Scientific Agents Transcriptomics Assay & Models ML-based Patient Connectivity (RWD) LLMs & Graphs MOA Deconvolution Scaled Binding Affinity Predictions ClinTech Workflow AI-enabled Precision Design AI-informed Clinical Development Scientific Agents Nomination Workflow Scientific Agents Supported by BioHive-2 The fastest, most powerful supercomputer wholly-owned and operated by a pharma or biotech company ~65PB of data including 40PB of proprietary data Phenomics Assay & Models (Early version) AI-powered Biological Insights
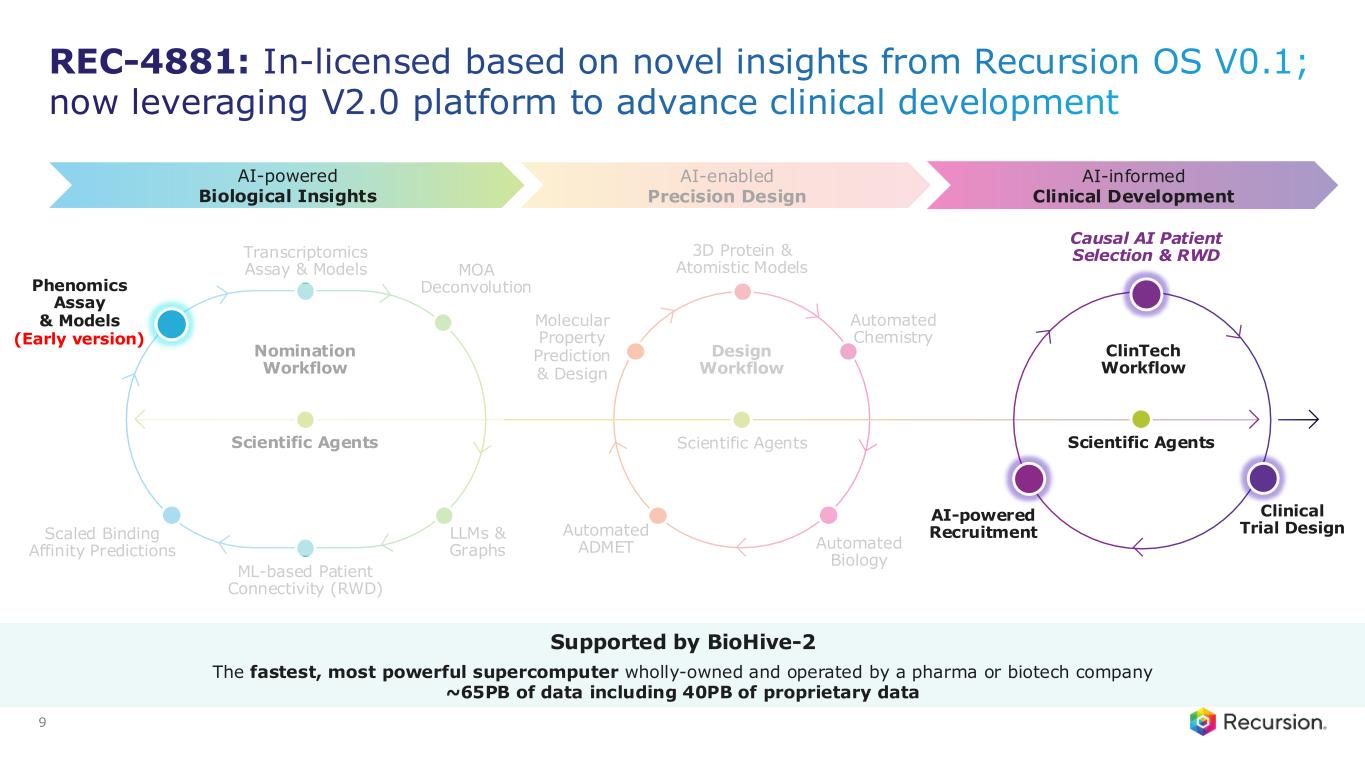
REC-4881: In-licensed based on novel insights from Recursion OS V0.1; now leveraging V2.0 platform to advance clinical development 9 Design Workflow Automated ADMET Automated Chemistry Automated Biology 3D Protein & Atomistic Models Molecular Property Prediction & Design Scientific Agents Transcriptomics Assay & Models ML-based Patient Connectivity (RWD) LLMs & Graphs MOA Deconvolution Scaled Binding Affinity Predictions AI-enabled Precision Design Clinical Trial Design ClinTech Workflow AI-powered Recruitment AI-informed Clinical Development Scientific Agents AI-powered Biological Insights Nomination Workflow Scientific Agents Supported by BioHive-2 The fastest, most powerful supercomputer wholly-owned and operated by a pharma or biotech company ~65PB of data including 40PB of proprietary data Causal AI Patient Selection & RWD Phenomics Assay & Models (Early version)
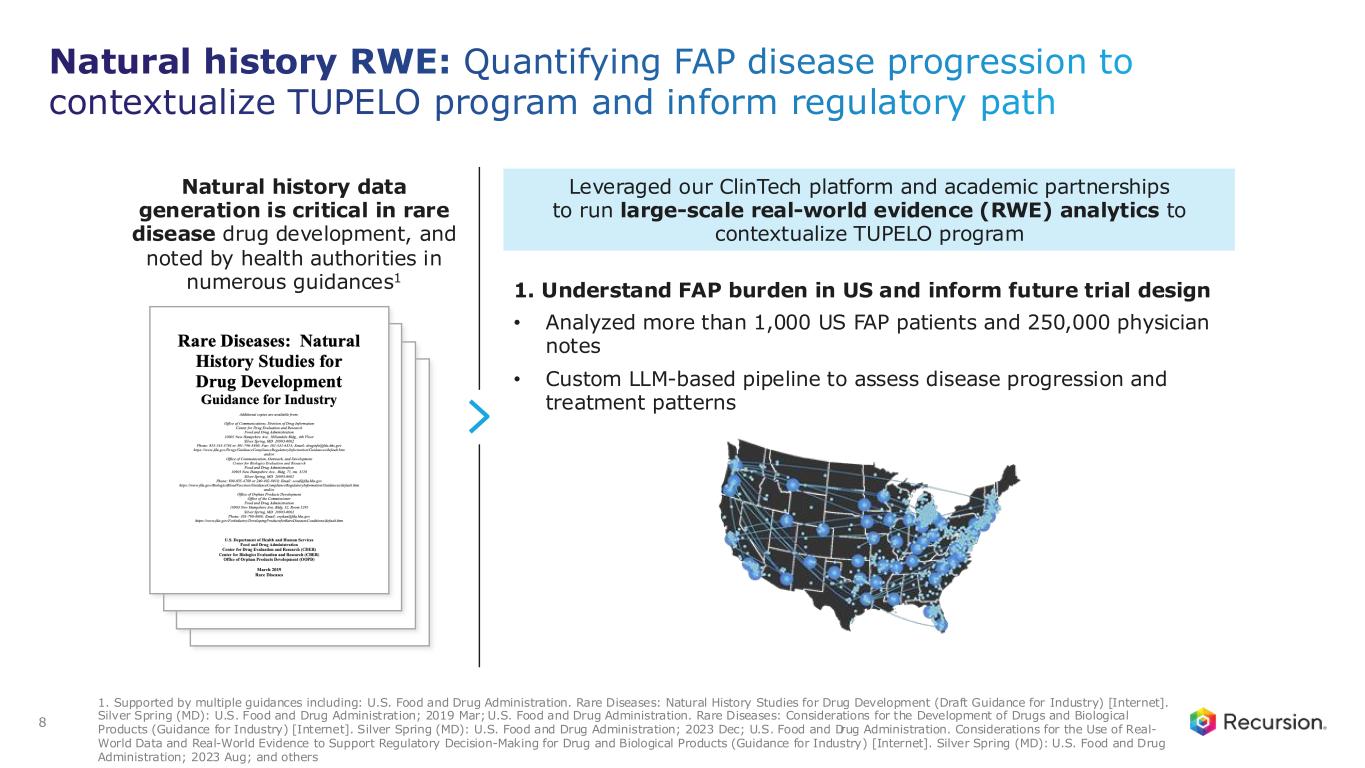
8 Natural history RWE: Quantifying FAP disease progression to contextualize TUPELO program and inform regulatory path 1. Supported by multiple guidances including: U.S. Food and Drug Administration. Rare Diseases: Natural History Studies for Drug Development (Draft Guidance for Industry) [Internet]. Silver Spring (MD): U.S. Food and Drug Administration; 2019 Mar; U.S. Food and Drug Administration. Rare Diseases: Considerations for the Development of Drugs and Biological Products (Guidance for Industry) [Internet]. Silver Spring (MD): U.S. Food and Drug Administration; 2023 Dec; U.S. Food and Drug Administration. Considerations for the Use of Real- World Data and Real-World Evidence to Support Regulatory Decision-Making for Drug and Biological Products (Guidance for Industry) [Internet]. Silver Spring (MD): U.S. Food and Drug Administration; 2023 Aug; and others Leveraged our ClinTech platform and academic partnerships to run large-scale real-world evidence (RWE) analytics to contextualize TUPELO program 1. Understand FAP burden in US and inform future trial design • Analyzed more than 1,000 US FAP patients and 250,000 physician notes • Custom LLM-based pipeline to assess disease progression and treatment patterns Natural history data generation is critical in rare disease drug development, and noted by health authorities in numerous guidances1
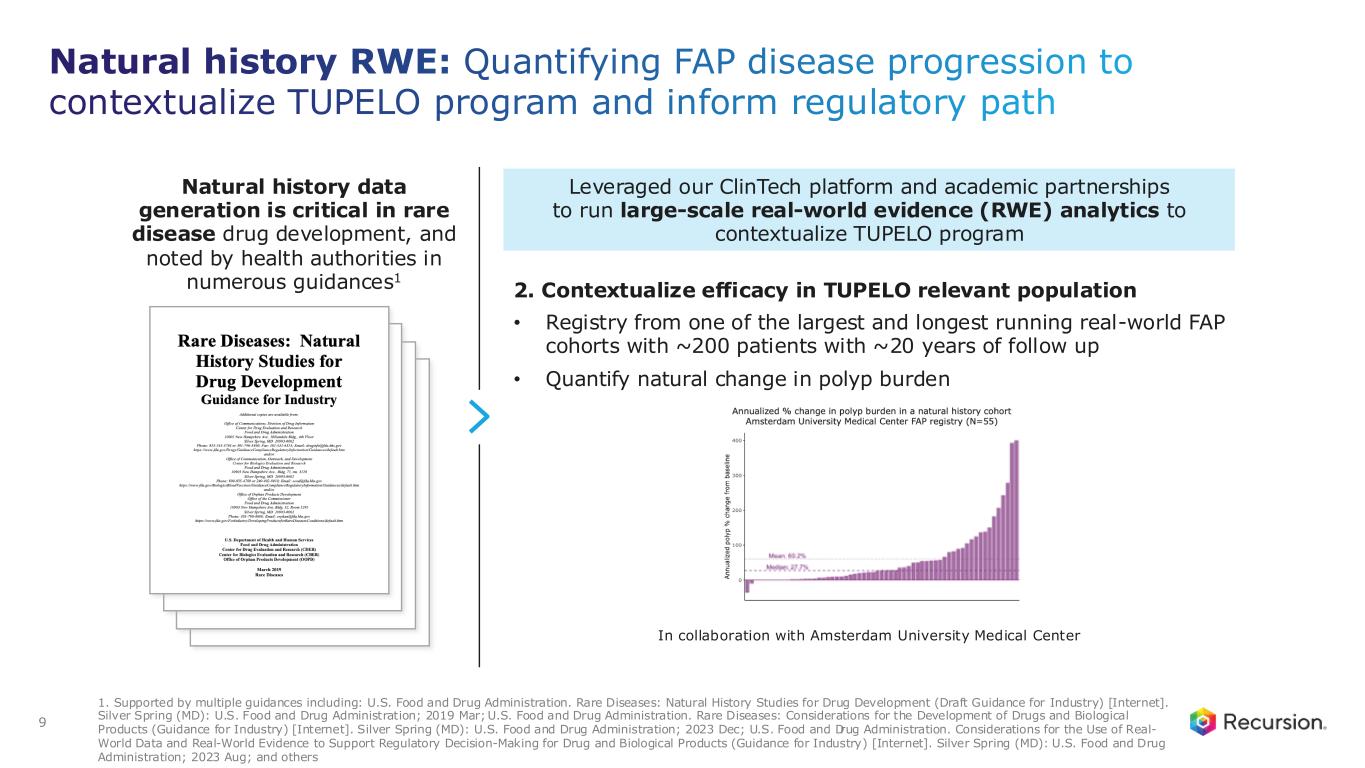
9 Natural history RWE: Quantifying FAP disease progression to contextualize TUPELO program and inform regulatory path 1. Supported by multiple guidances including: U.S. Food and Drug Administration. Rare Diseases: Natural History Studies for Drug Development (Draft Guidance for Industry) [Internet]. Silver Spring (MD): U.S. Food and Drug Administration; 2019 Mar; U.S. Food and Drug Administration. Rare Diseases: Considerations for the Development of Drugs and Biological Products (Guidance for Industry) [Internet]. Silver Spring (MD): U.S. Food and Drug Administration; 2023 Dec; U.S. Food and Drug Administration. Considerations for the Use of Real- World Data and Real-World Evidence to Support Regulatory Decision-Making for Drug and Biological Products (Guidance for Industry) [Internet]. Silver Spring (MD): U.S. Food and Drug Administration; 2023 Aug; and others Leveraged our ClinTech platform and academic partnerships to run large-scale real-world evidence (RWE) analytics to contextualize TUPELO program Natural history data generation is critical in rare disease drug development, and noted by health authorities in numerous guidances1 In collaboration with Amsterdam University Medical Center 2. Contextualize efficacy in TUPELO relevant population • Registry from one of the largest and longest running real-world FAP cohorts with ~200 patients with ~20 years of follow up • Quantify natural change in polyp burden
Fireside chat: Today’s speakers 10 Elizabeth Bruckheimer, PhD VP of Clinical Development Recursion • Highly accomplished executive with ≥20 years of industry experience, directing all phases of drug development from preclinical to clinical execution • Former VP and CSO Panbela Therapeutics / Cancer Prevention Pharmaceuticals leading largest FAP Phase 3 trial David Mauro, MD, PhD Chief Medical Officer Recursion • Former CMO Codiak BioSciences & Checkmate Pharmaceuticals • Former ED of Oncology Merck • Over 20 years of experience in oncology drug development, incl. spearheading early- and late-stage development for more than 25 INDs over the past decade Jessica Stout, DO Assistant Clinical Professor University of Utah School of Medicine • Double board-certified in Internal Medicine and Gastroenterology • Specializes in complex gastrointestinal disease management incl. long-term, multi-organ management of FAP Alfred M. Cohen, MD, FACS, FASCRS Former Chief of Colorectal Service Memorial Sloan-Kettering Cancer Center • Former CMO Cancer Prevention Pharmaceuticals • Former Director, CEO at Lucille P. Markey Cancer Center at University of Kentucky • >50 years of experience in CRC treatment & research, with clinical focus on QoL in patients with CRC, Lynch syndrome, and FAP

REC-4881 Program Elizabeth Bruckheimer, PhD | VP, Clinical Development

Disease context: FAP is a rare disease characterized by extensive polyps in the GI tract, with no approved pharmacotherapies • Orphan disease caused by autosomal dominant inactivating mutations in APC1 • One of the most clinically significant hereditary colorectal cancer syndromes • Majority of patients will develop 100s-1000s of colorectal adenomas with near- 100% CRC risk by age ~40 in absence of surgery or excisions • Adenomas progressively accumulate with limited evidence of spontaneous regression, driving disease burden throughout life • Lifetime of endoscopic surveillance, frequent excisional interventions, life-altering surgeries, and poor QoL, morbidity and mortality Disease Background High Unmet Need • ~50,000 addressable patients in US + EU52 • Surgery as the only standard-of-care • Current medical therapies have no impact on slowing disease progression • REC-4881 may be positioned to fill a significant unmet need with no approved pharmacotherapies 12 1. 20-30% of FAP patients have de novo mutations with no family history. Half E et al, Orphanet J Rar Dis. 2009 2. Internal company estimates Scope view source: Samir at the English-language Wikipedia, CC BY-SA 3.0, via Wikimedia Commons Polyps on mucosal membrane of colon Colonoscope Scope view
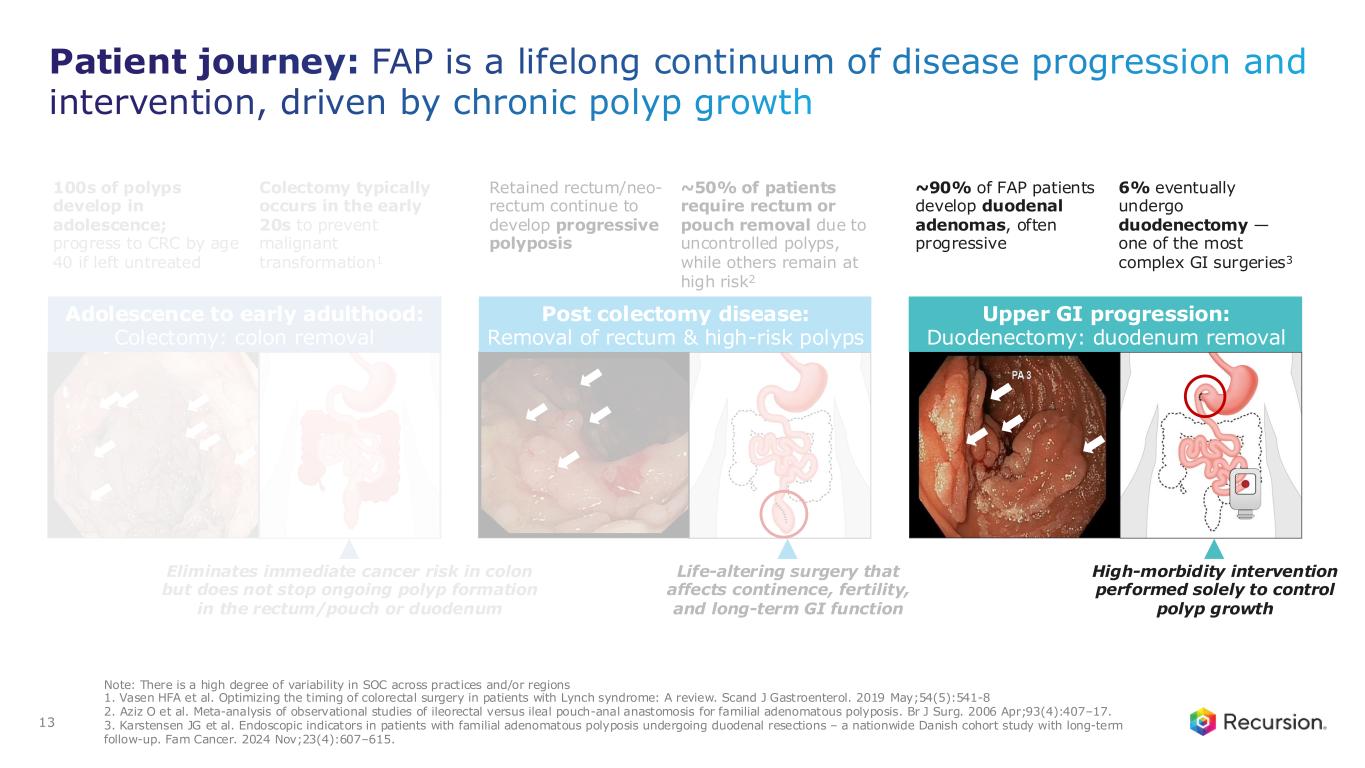
Patient journey: FAP is a lifelong continuum of disease progression and intervention, driven by chronic polyp growth 13 Note: There is a high degree of variability in SOC across practices and/or regions 1. Vasen HFA et al. Optimizing the timing of colorectal surgery in patients with Lynch syndrome: A review. Scand J Gastroenterol. 2019 May;54(5):541-8 2. Aziz O et al. Meta-analysis of observational studies of ileorectal versus ileal pouch-anal anastomosis for familial adenomatous polyposis. Br J Surg. 2006 Apr;93(4):407–17. 3. Karstensen JG et al. Endoscopic indicators in patients with familial adenomatous polyposis undergoing duodenal resections – a nationwide Danish cohort study with long-term follow-up. Fam Cancer. 2024 Nov;23(4):607–615. 100s of polyps develop in adolescence; progress to CRC by age 40 if left untreated Colectomy typically occurs in the early 20s to prevent malignant transformation1 Adolescence to early adulthood: Colectomy: colon removal Eliminates immediate cancer risk in colon but does not stop ongoing polyp formation in the rectum/pouch or duodenum Retained rectum/neo- rectum continue to develop progressive polyposis ~50% of patients require rectum or pouch removal due to uncontrolled polyps, while others remain at high risk2 Post colectomy disease: Removal of rectum & high-risk polyps Life-altering surgery that affects continence, fertility, and long-term GI function ~90% of FAP patients develop duodenal adenomas, often progressive 6% eventually undergo duodenectomy — one of the most complex GI surgeries3 Upper GI progression: Duodenectomy: duodenum removal High-morbidity intervention performed solely to control polyp growth

ClinTech insights: Real-world US electronic health records and physician notes highlight disease progression in FAP patients Note: all reported rates are adjusted for variable follow up time. Data analysis includes unstructured physician notes and structured components of EHRs such as billing codes for phenotype definitions. 1. Notes were processed using a custom NLP pipeline with Recursion-built and operated supercomputer, BioHive-2 2. Polypectomy rates are expected to be under-estimated due to documentation practices in routine clinical care 3. Amoyel M, Belle A, Dhooge M, et al. Outcomes of endoscopic mucosal resection for large superficial non-ampullary duodenal adenomas. Sci Rep. 2022;12:14592. doi:10.1038/s41598-022-18528-7 4. Lemos Garcia J, Rosa I, Pereira da Silva J, Lage P, Claro I. Endoscopic Approach to Duodenal Adenomas in Familial Adenomatous Polyposis: A Retrospective Cohort. GE Port J Gastroenterol. 2023;30:430-436. doi:10.1159/000527209 5. Major surgeries were defined as ileal pouch-anal anastomosis, colectomy, proctocolectomy, Whipple, ileostomy, and ampullectomy. Analysis captures documentation of “history of” such procedures. Major surgeries are expected to be underestimated due to limited follow up period and potential underreporting of patient medical history. 14 • Patients commonly had disease progression and continuous intervention • Limited evidence of spontaneous polyp burden reduction in any FAP patient • Frequent polypectomies with at least 5 polyps removed per patient per year2 • Advanced excisions are associated with risk of complications such as perforation and bleeding3,4 • At least 75% of patients had a major FAP-related surgery documented5 1,068 FAP patients and 256,000 physician notes were analyzed during period of 2017-20251 “No polyps were removed given there were an innumerable amount (>300) and removal would not be endoscopically feasible." “[they] had greater than 10 surgeries and multiple revisions of [their] colectomy and resection of [their] small bowel.” “[they] underwent a Whipple procedure because the duodenal and ampullary polyp burden was too high.”
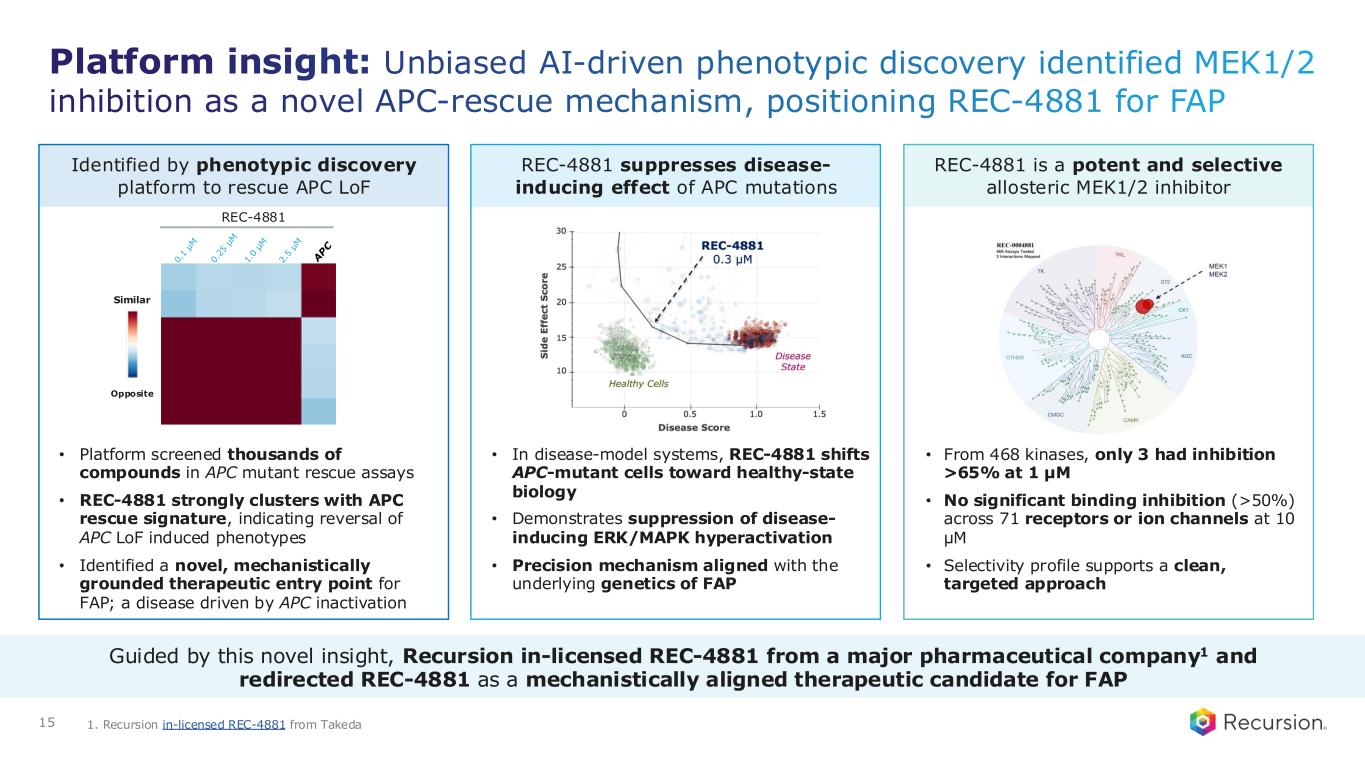
Platform insight: Unbiased AI-driven phenotypic discovery identified MEK1/2 inhibition as a novel APC-rescue mechanism, positioning REC-4881 for FAP • Platform screened thousands of compounds in APC mutant rescue assays • REC-4881 strongly clusters with APC rescue signature, indicating reversal of APC LoF induced phenotypes • Identified a novel, mechanistically grounded therapeutic entry point for FAP; a disease driven by APC inactivation Identified by phenotypic discovery platform to rescue APC LoF • In disease-model systems, REC-4881 shifts APC-mutant cells toward healthy-state biology • Demonstrates suppression of disease- inducing ERK/MAPK hyperactivation • Precision mechanism aligned with the underlying genetics of FAP REC-4881 suppresses disease- inducing effect of APC mutations • From 468 kinases, only 3 had inhibition >65% at 1 µM • No significant binding inhibition (>50%) across 71 receptors or ion channels at 10 µM • Selectivity profile supports a clean, targeted approach REC-4881 is a potent and selective allosteric MEK1/2 inhibitor REC-4881 Similar Opposite 15 Guided by this novel insight, Recursion in-licensed REC-4881 from a major pharmaceutical company1 and redirected REC-4881 as a mechanistically aligned therapeutic candidate for FAP 1. Recursion in-licensed REC-4881 from Takeda
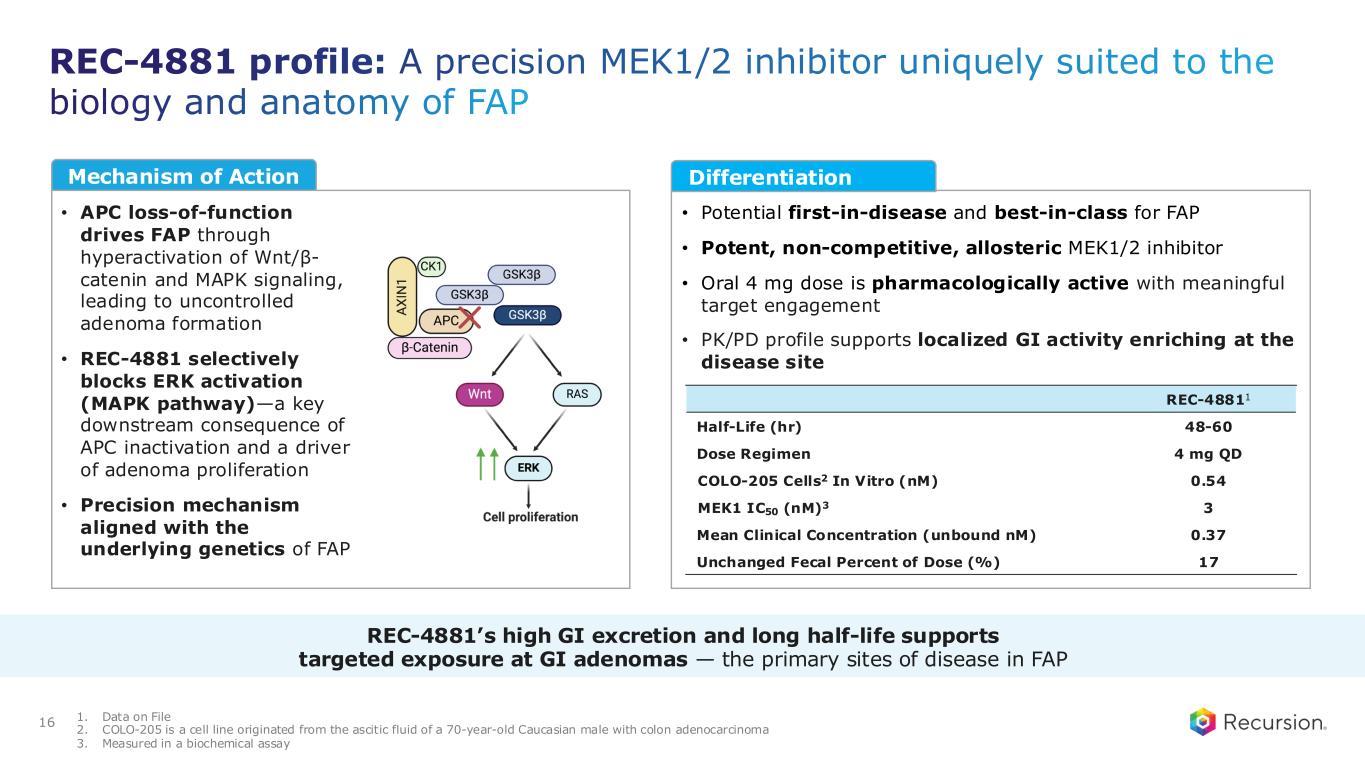
REC-4881 profile: A precision MEK1/2 inhibitor uniquely suited to the biology and anatomy of FAP 16 • APC loss-of-function drives FAP through hyperactivation of Wnt/β- catenin and MAPK signaling, leading to uncontrolled adenoma formation • REC-4881 selectively blocks ERK activation (MAPK pathway)—a key downstream consequence of APC inactivation and a driver of adenoma proliferation • Precision mechanism aligned with the underlying genetics of FAP • Potential first-in-disease and best-in-class for FAP • Potent, non-competitive, allosteric MEK1/2 inhibitor • Oral 4 mg dose is pharmacologically active with meaningful target engagement • PK/PD profile supports localized GI activity enriching at the disease site REC-48811 Half-Life (hr) 48-60 Dose Regimen 4 mg QD COLO-205 Cells2 In Vitro (nM) 0.54 MEK1 IC50 (nM)3 3 Mean Clinical Concentration (unbound nM) 0.37 Unchanged Fecal Percent of Dose (%) 17 1. Data on File 2. COLO-205 is a cell line originated from the ascitic fluid of a 70-year-old Caucasian male with colon adenocarcinoma 3. Measured in a biochemical assay REC-4881’s high GI excretion and long half-life supports targeted exposure at GI adenomas — the primary sites of disease in FAP Mechanism of Action Differentiation
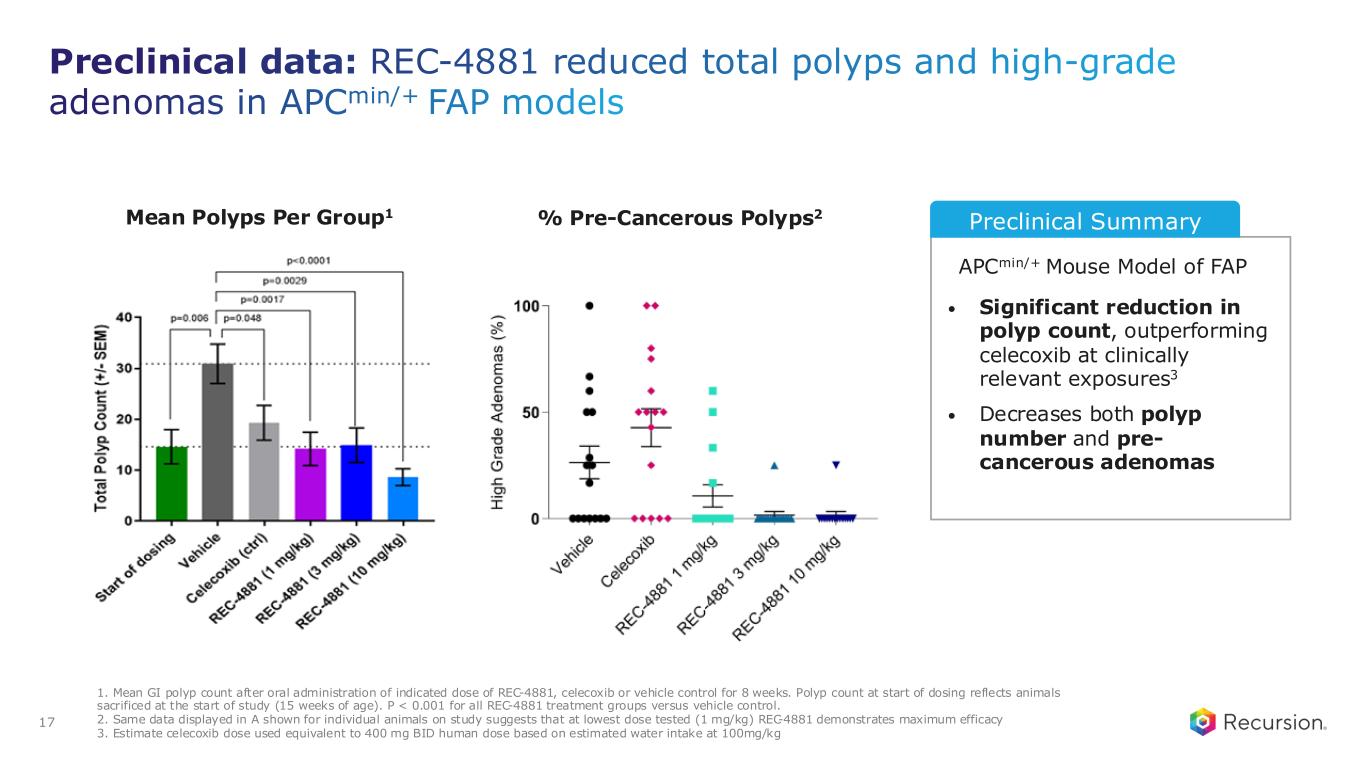
Preclinical data: REC-4881 reduced total polyps and high-grade adenomas in APCmin/+ FAP models APCmin/+ Mouse Model of FAP • Significant reduction in polyp count, outperforming celecoxib at clinically relevant exposures3 • Decreases both polyp number and pre- cancerous adenomas % Pre-Cancerous Polyps2 Preclinical Summary 1. Mean GI polyp count after oral administration of indicated dose of REC-4881, celecoxib or vehicle control for 8 weeks. Polyp count at start of dosing reflects animals sacrificed at the start of study (15 weeks of age). P < 0.001 for all REC-4881 treatment groups versus vehicle control. 2. Same data displayed in A shown for individual animals on study suggests that at lowest dose tested (1 mg/kg) REC-4881 demonstrates maximum efficacy 3. Estimate celecoxib dose used equivalent to 400 mg BID human dose based on estimated water intake at 100mg/kg 17 Mean Polyps Per Group1

Ongoing Phase 1b/2 Updated Results & Natural History Analysis
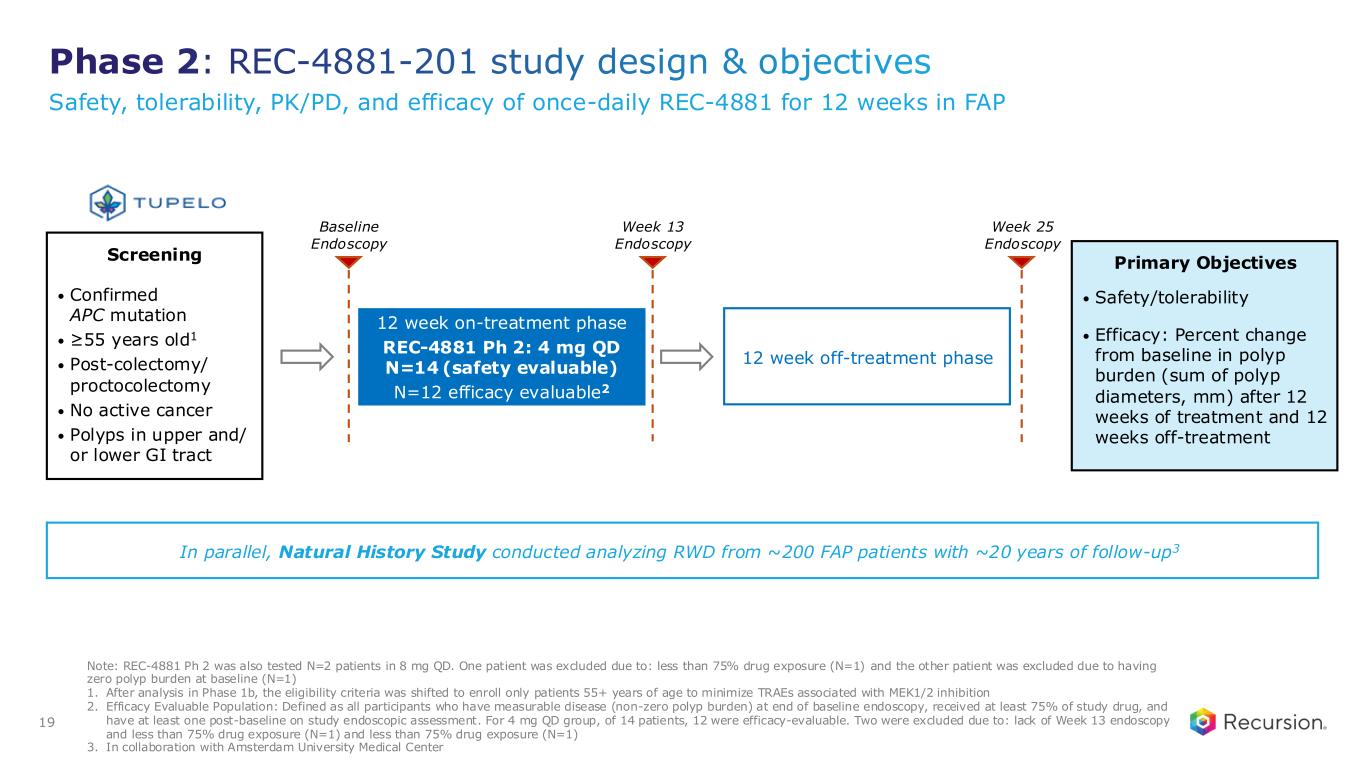
Phase 2: REC-4881-201 study design & objectives Note: REC-4881 Ph 2 was also tested N=2 patients in 8 mg QD. One patient was excluded due to: less than 75% drug exposure (N=1) and the other patient was excluded due to having zero polyp burden at baseline (N=1) 1. After analysis in Phase 1b, the eligibility criteria was shifted to enroll only patients 55+ years of age to minimize TRAEs associated with MEK1/2 inhibition 2. Efficacy Evaluable Population: Defined as all participants who have measurable disease (non-zero polyp burden) at end of baseline endoscopy, received at least 75% of study drug, and have at least one post-baseline on study endoscopic assessment. For 4 mg QD group, of 14 patients, 12 were efficacy-evaluable. Two were excluded due to: lack of Week 13 endoscopy and less than 75% drug exposure (N=1) and less than 75% drug exposure (N=1) 3. In collaboration with Amsterdam University Medical Center 19 In parallel, Natural History Study conducted analyzing RWD from ~200 FAP patients with ~20 years of follow-up3 Screening • Confirmed APC mutation • ≥55 years old1 • Post-colectomy/ proctocolectomy • No active cancer • Polyps in upper and/ or lower GI tract Primary Objectives • Safety/tolerability • Efficacy: Percent change from baseline in polyp burden (sum of polyp diameters, mm) after 12 weeks of treatment and 12 weeks off-treatment 12 week off-treatment phase 12 week on-treatment phase REC-4881 Ph 2: 4 mg QD N=14 (safety evaluable) N=12 efficacy evaluable2 Week 13 Endoscopy Baseline Endoscopy Week 25 Endoscopy Safety, tolerability, PK/PD, and efficacy of once-daily REC-4881 for 12 weeks in FAP
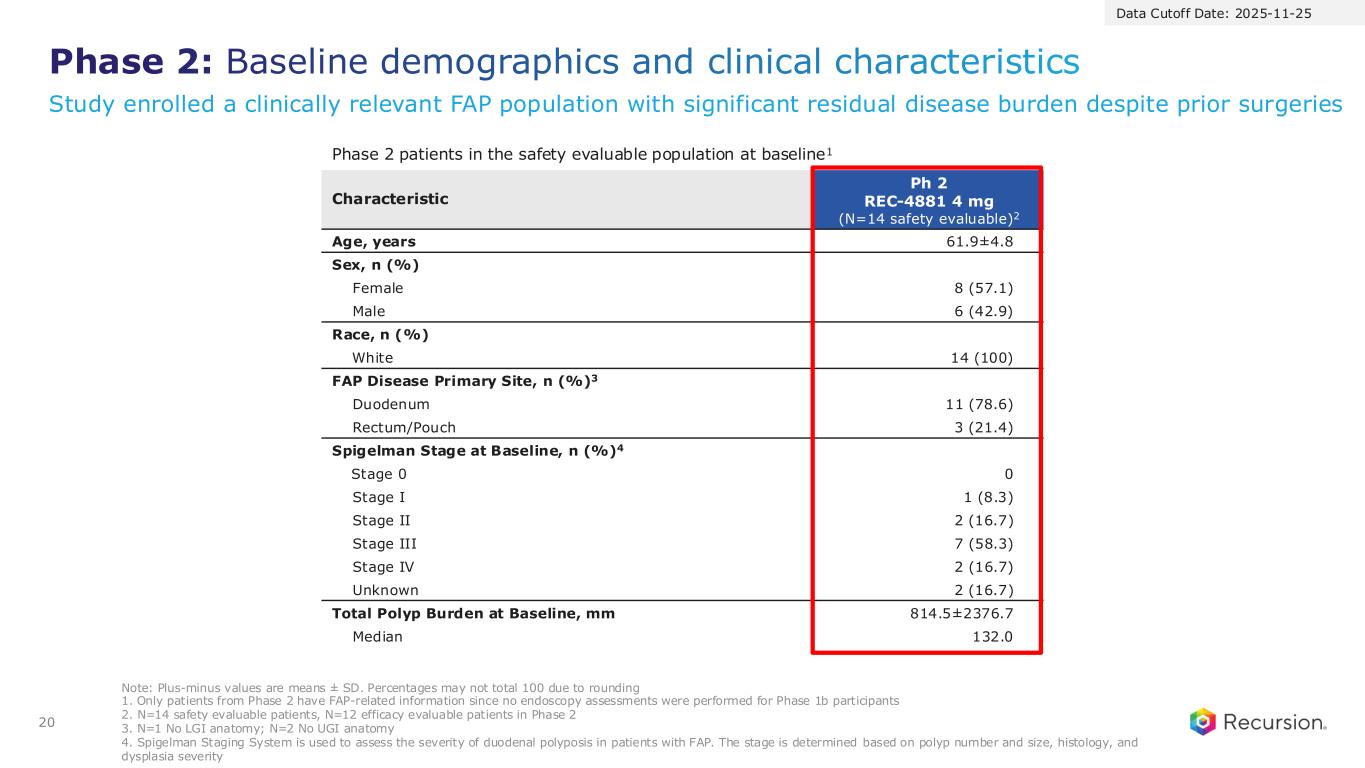
Phase 2: Baseline demographics and clinical characteristics 20 Characteristic Ph 2 REC-4881 4 mg (N=14 safety evaluable)2 Age, years 61.9±4.8 Sex, n (%) Female 8 (57.1) Male 6 (42.9) Race, n (%) White 14 (100) FAP Disease Primary Site, n (%)3 Duodenum 11 (78.6) Rectum/Pouch 3 (21.4) Spigelman Stage at Baseline, n (%)4 Stage 0 0 Stage I 1 (8.3) Stage II 2 (16.7) Stage III 7 (58.3) Stage IV 2 (16.7) Unknown 2 (16.7) Total Polyp Burden at Baseline, mm 814.5±2376.7 Median 132.0 Note: Plus-minus values are means ± SD. Percentages may not total 100 due to rounding 1. Only patients from Phase 2 have FAP-related information since no endoscopy assessments were performed for Phase 1b participants 2. N=14 safety evaluable patients, N=12 efficacy evaluable patients in Phase 2 3. N=1 No LGI anatomy; N=2 No UGI anatomy 4. Spigelman Staging System is used to assess the severity of duodenal polyposis in patients with FAP. The stage is determined based on polyp number and size, histology, and dysplasia severity Phase 2 patients in the safety evaluable population at baseline1 Data Cutoff Date: 2025-11-25 Study enrolled a clinically relevant FAP population with significant residual disease burden despite prior surgeries
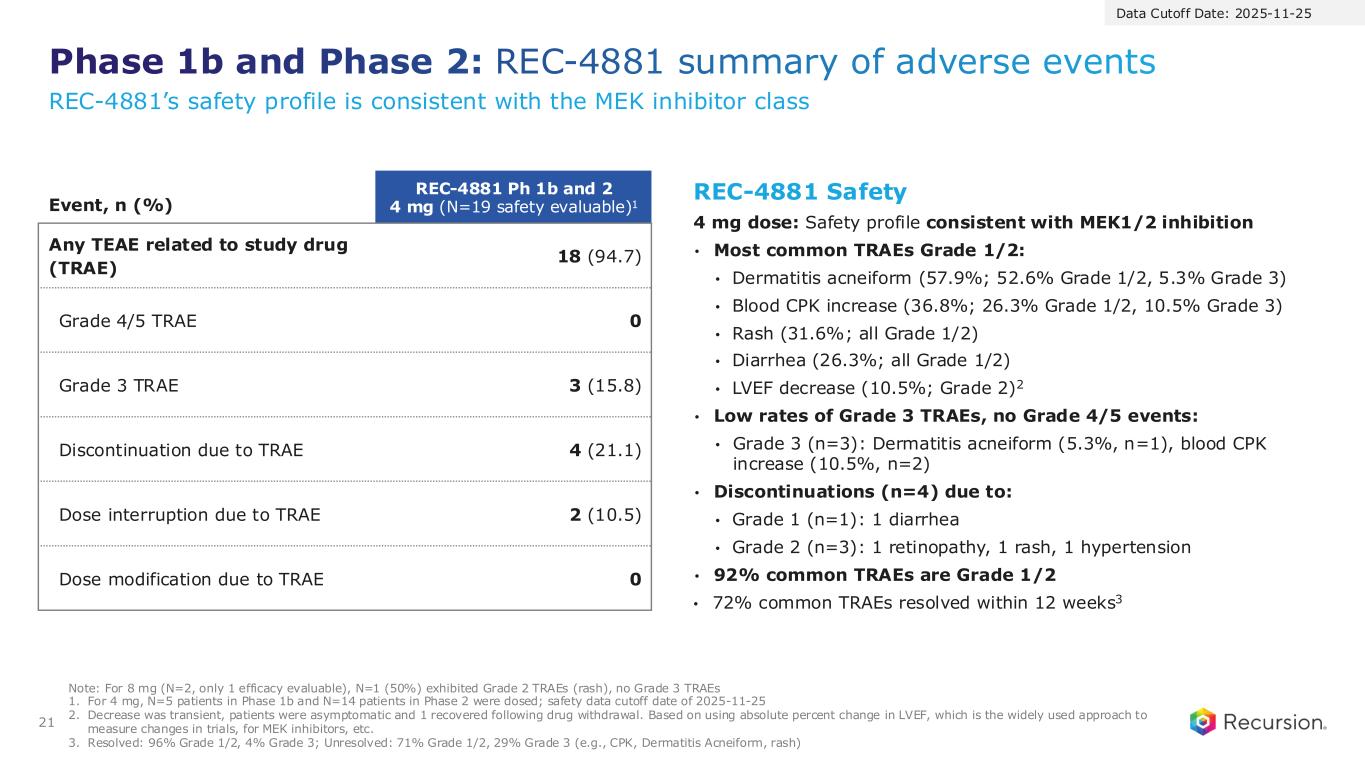
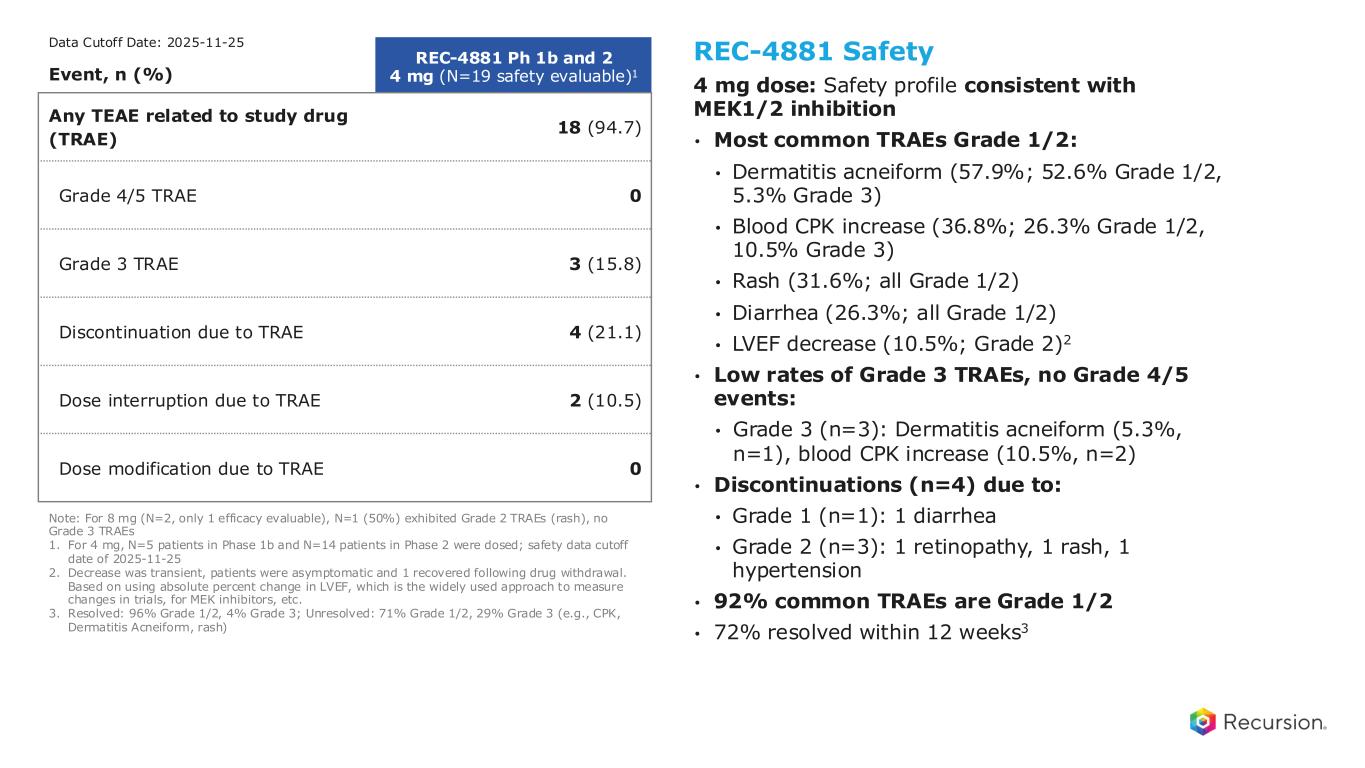
Phase 1b and Phase 2: REC-4881 summary of adverse events 21 Note: For 8 mg (N=2, only 1 efficacy evaluable), N=1 (50%) exhibited Grade 2 TRAEs (rash), no Grade 3 TRAEs 1. For 4 mg, N=5 patients in Phase 1b and N=14 patients in Phase 2 were dosed; safety data cutoff date of 2025-11-25 2. Decrease was transient, patients were asymptomatic and 1 recovered following drug withdrawal. Based on using absolute percent change in LVEF, which is the widely used approach to measure changes in trials, for MEK inhibitors, etc. 3. Resolved: 96% Grade 1/2, 4% Grade 3; Unresolved: 71% Grade 1/2, 29% Grade 3 (e.g., CPK, Dermatitis Acneiform, rash) Event, n (%) REC-4881 Ph 1b and 2 4 mg (N=19 safety evaluable)1 Any TEAE related to study drug (TRAE) 18 (94.7) Grade 4/5 TRAE 0 Grade 3 TRAE 3 (15.8) Discontinuation due to TRAE 4 (21.1) Dose interruption due to TRAE 2 (10.5) Dose modification due to TRAE 0 REC-4881 Safety 4 mg dose: Safety profile consistent with MEK1/2 inhibition • Most common TRAEs Grade 1/2: • Dermatitis acneiform (57.9%; 52.6% Grade 1/2, 5.3% Grade 3) • Blood CPK increase (36.8%; 26.3% Grade 1/2, 10.5% Grade 3) • Rash (31.6%; all Grade 1/2) • Diarrhea (26.3%; all Grade 1/2) • LVEF decrease (10.5%; Grade 2)2 • Low rates of Grade 3 TRAEs, no Grade 4/5 events: • Grade 3 (n=3): Dermatitis acneiform (5.3%, n=1), blood CPK increase (10.5%, n=2) • Discontinuations (n=4) due to: • Grade 1 (n=1): 1 diarrhea • Grade 2 (n=3): 1 retinopathy, 1 rash, 1 hypertension • 92% common TRAEs are Grade 1/2 • 72% common TRAEs resolved within 12 weeks3 Data Cutoff Date: 2025-11-25 REC-4881’s safety profile is consistent with the MEK inhibitor class
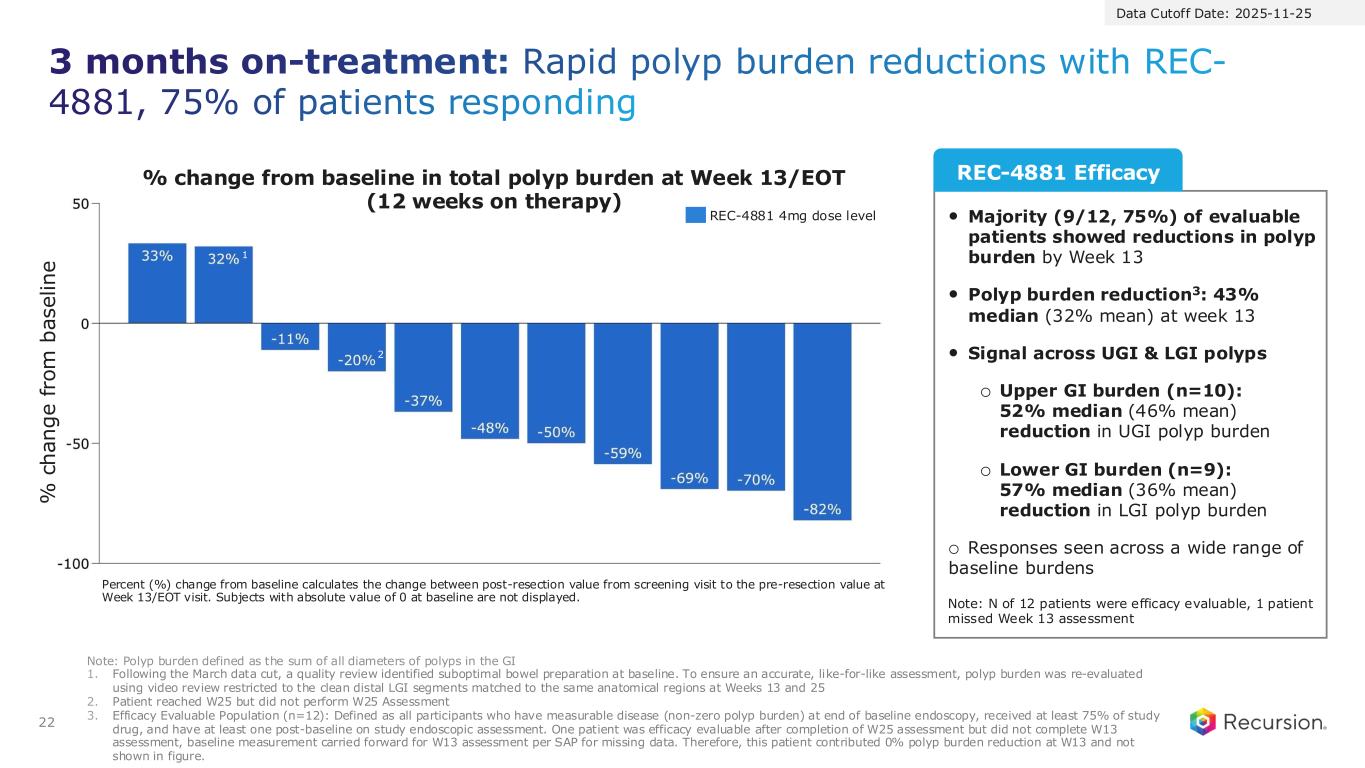
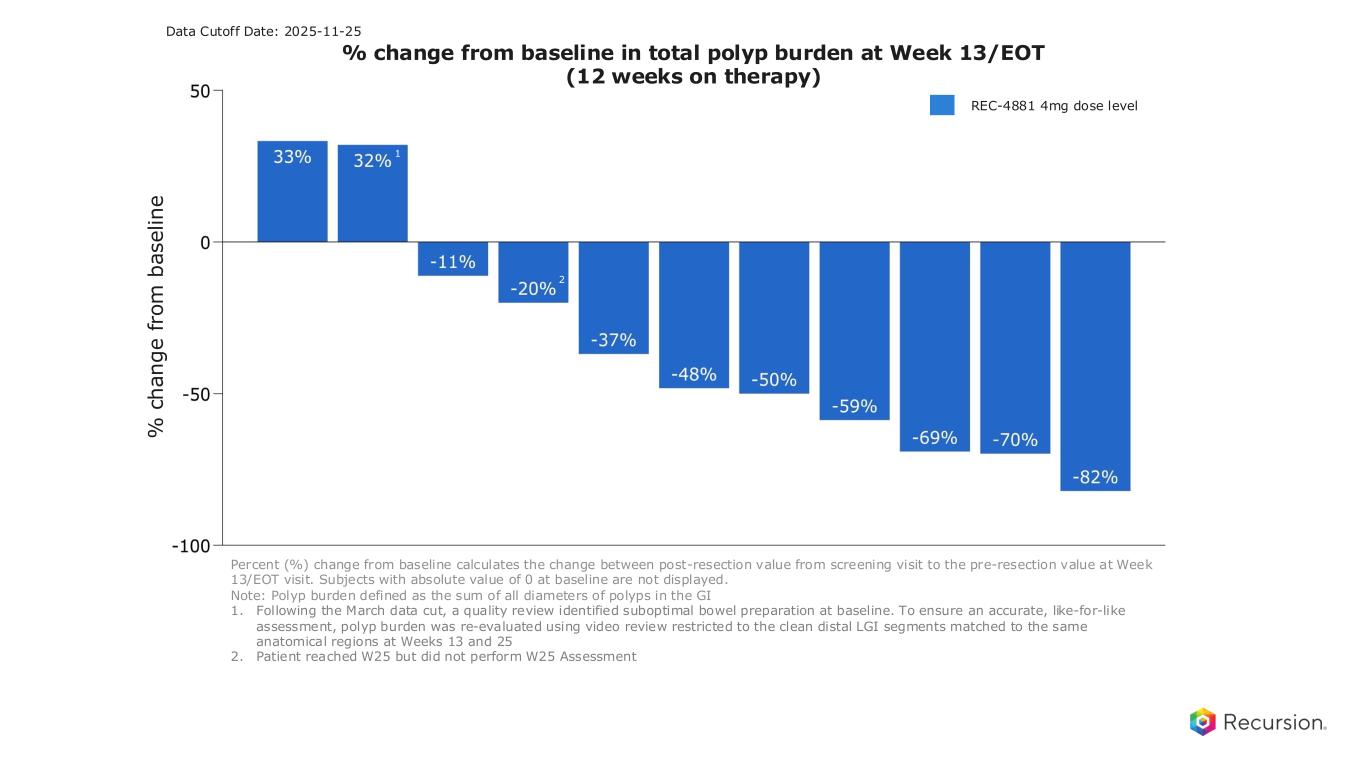
22 3 months on-treatment: Rapid polyp burden reductions with REC- 4881, 75% of patients responding Percent (%) change from baseline calculates the change between post-resection value from screening visit to the pre-resection value at Week 13/EOT visit. Subjects with absolute value of 0 at baseline are not displayed. Note: Polyp burden defined as the sum of all diameters of polyps in the GI 1. Following the March data cut, a quality review identified suboptimal bowel preparation at baseline. To ensure an accurate, like-for-like assessment, polyp burden was re-evaluated using video review restricted to the clean distal LGI segments matched to the same anatomical regions at Weeks 13 and 25 2. Patient reached W25 but did not perform W25 Assessment 3. Efficacy Evaluable Population (n=12): Defined as all participants who have measurable disease (non-zero polyp burden) at end of baseline endoscopy, received at least 75% of study drug, and have at least one post-baseline on study endoscopic assessment. One patient was efficacy evaluable after completion of W25 assessment but did not complete W13 assessment, baseline measurement carried forward for W13 assessment per SAP for missing data. Therefore, this patient contributed 0% polyp burden reduction at W13 and not shown in figure. • Majority (9/12, 75%) of evaluable patients showed reductions in polyp burden by Week 13 • Polyp burden reduction3: 43% median (32% mean) at week 13 • Signal across UGI & LGI polyps o Upper GI burden (n=10): 52% median (46% mean) reduction in UGI polyp burden o Lower GI burden (n=9): 57% median (36% mean) reduction in LGI polyp burden o Responses seen across a wide range of baseline burdens Note: N of 12 patients were efficacy evaluable, 1 patient missed Week 13 assessment REC-4881 Efficacy REC-4881 4mg dose level % change from baseline in total polyp burden at Week 13/EOT (12 weeks on therapy) % c h a n g e f ro m b a s e li n e Data Cutoff Date: 2025-11-25 1 2
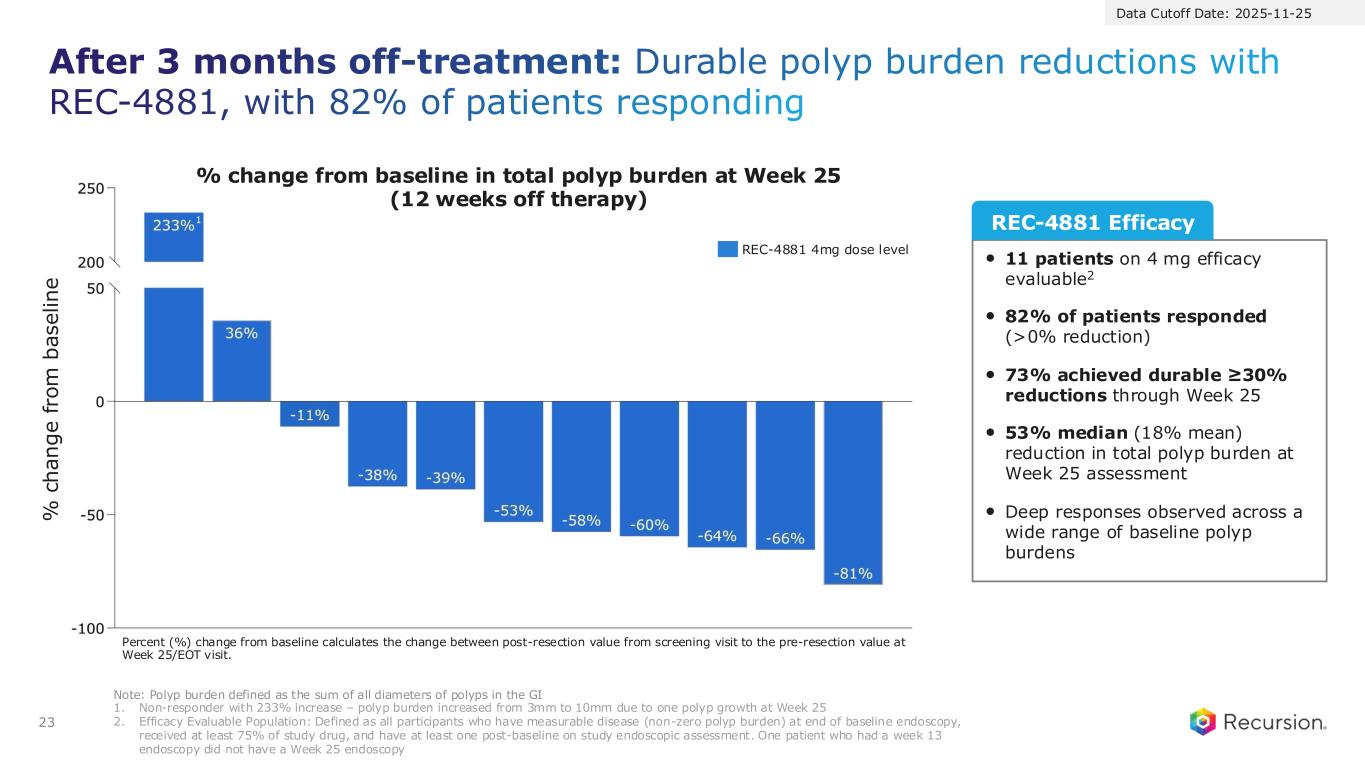
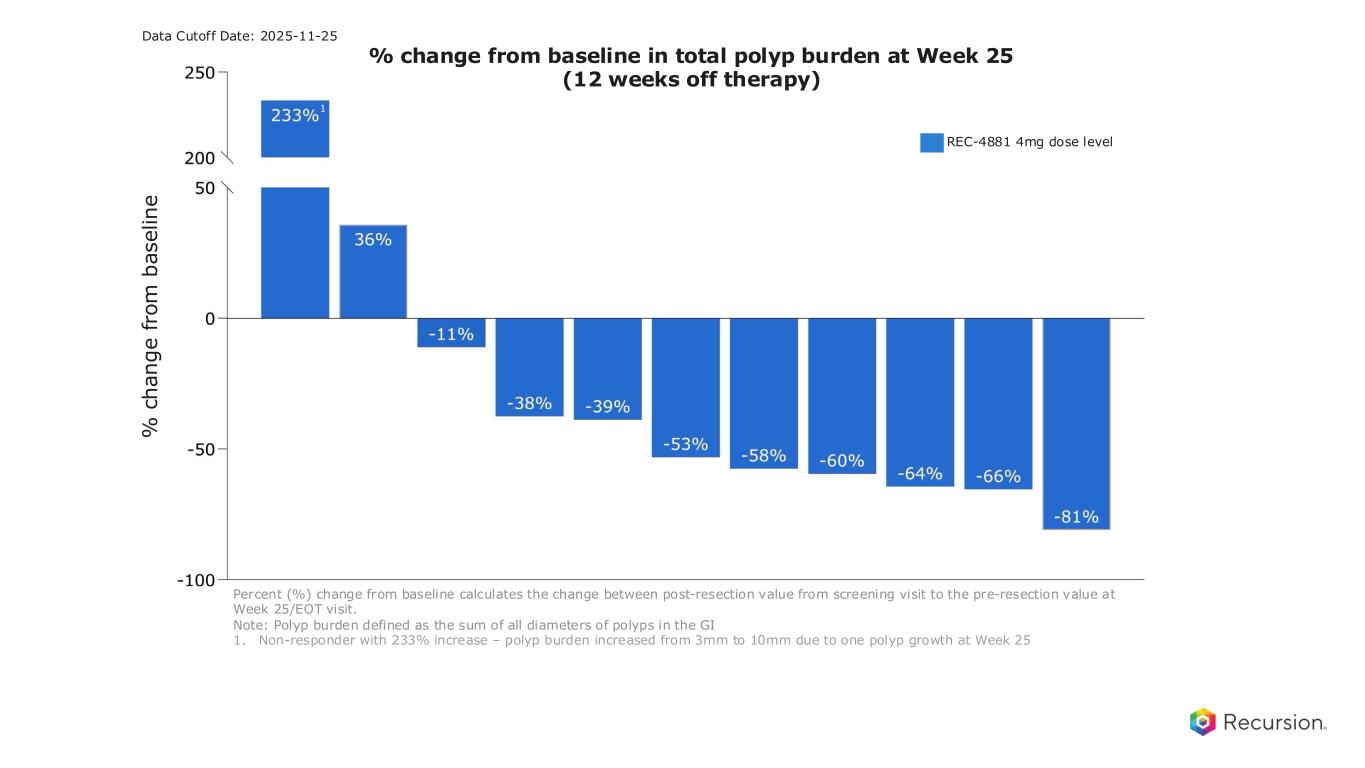
23 After 3 months off-treatment: Durable polyp burden reductions with REC-4881, with 82% of patients responding Note: Polyp burden defined as the sum of all diameters of polyps in the GI 1. Non-responder with 233% increase – polyp burden increased from 3mm to 10mm due to one polyp growth at Week 25 2. Efficacy Evaluable Population: Defined as all participants who have measurable disease (non-zero polyp burden) at end of baseline endoscopy, received at least 75% of study drug, and have at least one post-baseline on study endoscopic assessment. One patient who had a week 13 endoscopy did not have a Week 25 endoscopy • 11 patients on 4 mg efficacy evaluable2 • 82% of patients responded (>0% reduction) • 73% achieved durable ≥30% reductions through Week 25 • 53% median (18% mean) reduction in total polyp burden at Week 25 assessment • Deep responses observed across a wide range of baseline polyp burdens REC-4881 Efficacy Data Cutoff Date: 2025-11-25 REC-4881 4mg dose level % change from baseline in total polyp burden at Week 25 (12 weeks off therapy) % c h a n g e f ro m b a s e li n e Percent (%) change from baseline calculates the change between post-resection value from screening visit to the pre-resection value at Week 25/EOT visit. 1
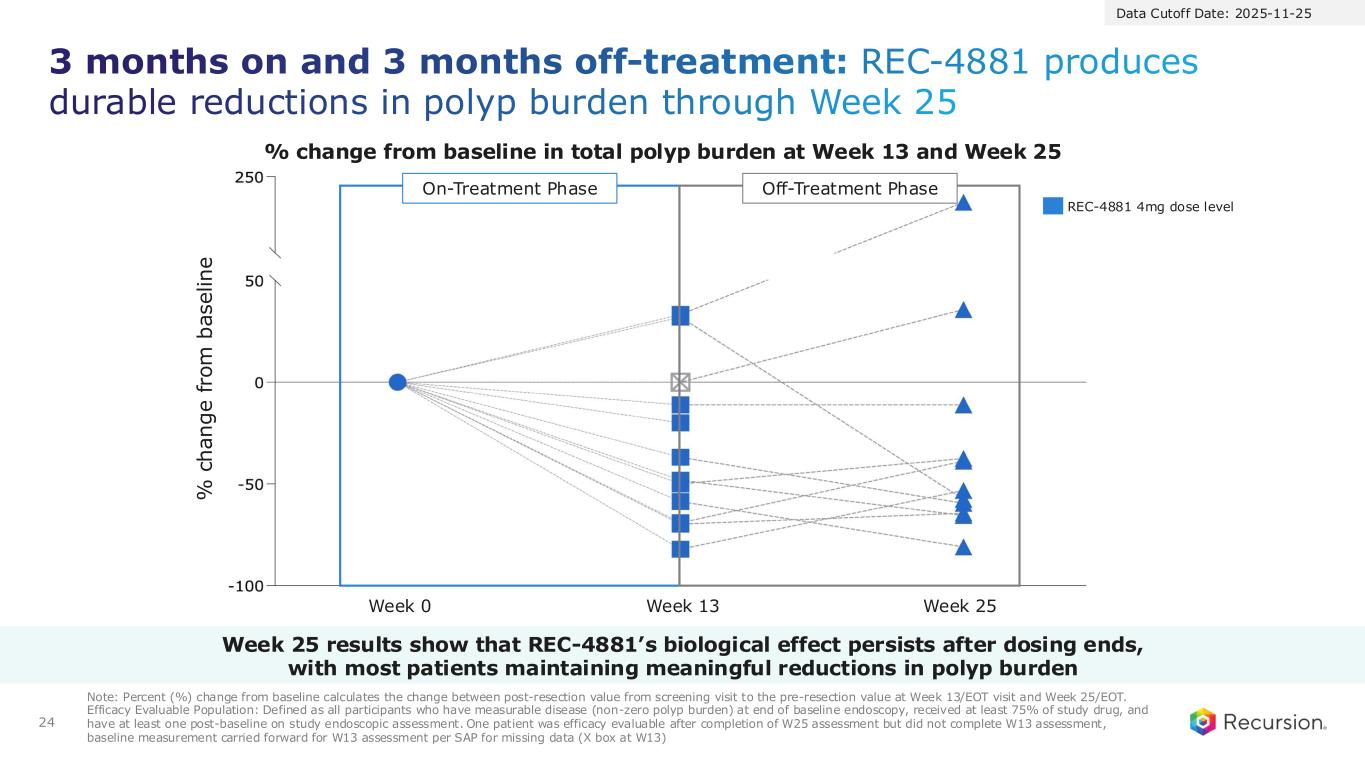
24 3 months on and 3 months off-treatment: REC-4881 produces durable reductions in polyp burden through Week 25 Note: Percent (%) change from baseline calculates the change between post-resection value from screening visit to the pre-resection value at Week 13/EOT visit and Week 25/EOT. Efficacy Evaluable Population: Defined as all participants who have measurable disease (non-zero polyp burden) at end of baseline endoscopy, received at least 75% of study drug, and have at least one post-baseline on study endoscopic assessment. One patient was efficacy evaluable after completion of W25 assessment but did not complete W13 assessment, baseline measurement carried forward for W13 assessment per SAP for missing data (X box at W13) REC-4881 4mg dose level % change from baseline in total polyp burden at Week 13 and Week 25 % c h a n g e f ro m b a s e li n e Week 0 Week 13 Week 25 On-Treatment Phase Off-Treatment Phase Data Cutoff Date: 2025-11-25 Week 25 results show that REC-4881’s biological effect persists after dosing ends, with most patients maintaining meaningful reductions in polyp burden
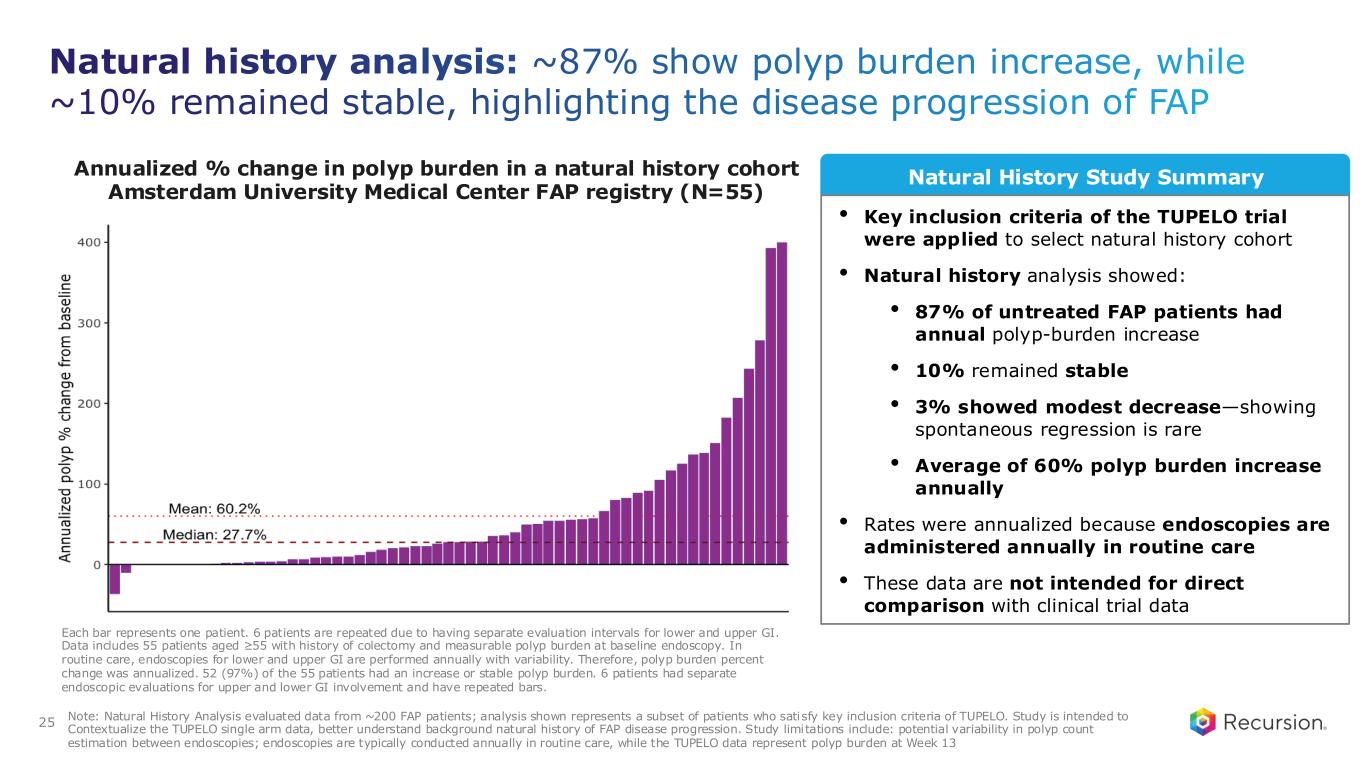
Natural history analysis: ~87% show polyp burden increase, while ~10% remained stable, highlighting the disease progression of FAP 25 Annualized % change in polyp burden in a natural history cohort Amsterdam University Medical Center FAP registry (N=55) Natural History Study Summary Each bar represents one patient. 6 patients are repeated due to having separate evaluation intervals for lower and upper GI. Data includes 55 patients aged ≥55 with history of colectomy and measurable polyp burden at baseline endoscopy. In routine care, endoscopies for lower and upper GI are performed annually with variability. Therefore, polyp burden percent change was annualized. 52 (97%) of the 55 patients had an increase or stable polyp burden. 6 patients had separate endoscopic evaluations for upper and lower GI involvement and have repeated bars. Note: Natural History Analysis evaluated data from ~200 FAP patients; analysis shown represents a subset of patients who satisfy key inclusion criteria of TUPELO. Study is intended to Contextualize the TUPELO single arm data, better understand background natural history of FAP disease progression. Study limitations include: potential variability in polyp count estimation between endoscopies; endoscopies are typically conducted annually in routine care, while the TUPELO data represent polyp burden at Week 13 • Key inclusion criteria of the TUPELO trial were applied to select natural history cohort • Natural history analysis showed: • 87% of untreated FAP patients had annual polyp-burden increase • 10% remained stable • 3% showed modest decrease—showing spontaneous regression is rare • Average of 60% polyp burden increase annually • Rates were annualized because endoscopies are administered annually in routine care • These data are not intended for direct comparison with clinical trial data

Week 13 Spigelman stage: REC-4881 produced Spigelman downstaging in 40% of patients, reflecting UGI disease improvement 26 AUMC: Amsterdam University Medical Center Note: N=10 efficacy evaluable for UGI; 2 patients lacked UGI anatomy. Spigelman Stage can be confounded by sampling errors 1. Patient did not perform WK13 endoscopy and so no biopsy collected 2. No biopsy sample collected at WK25 to calculate Spigelman Score Patient Spigelman Stage Initial screening –> WK13 –> WK25 1 III -> III -> III 2 I -> NA1 -> II 3 III -> II -> II 4 II -> II -> II 5 III -> II -> II 6 IV -> IV -> IV 7 III -> III -> III 8 III -> III -> NA2 8 III -> I -> I 10 IV -> II -> III Improvements reflect concordant reductions in the upper GI including polyp burden reduction, polyp count, and Spigelman downstaging • WK13: • 4/10 (40%) patients achieved a ≥1-point change in Spigelman Stage • Spigelman downstaging reflects improvement in duodenal polyposis severity — a major unmet need with limited therapeutic options. • WK25: • 4/10 patients maintained a ≥1-point change in Spigelman Stage • 1 patient had a 1-point increase • 1 patient did not have Week 25 endoscopy REC-4881 Efficacy Data Cutoff Date: 2025-11-25 Natural history RWD analysis showed 20% of patients experienced an increase in Spigelman stage between annual endoscopies in routine care. No decrease in Spigelman stage was observed.

Summary and Next Steps

REC-4881: Phase 1b/2 data review and next steps (I/II) 28 REC-4881 Potential First-in-Disease and Best-in-Class First Clinical Validation for Recursion OS Platform Platform-Generated Novel Mechanistic Insight → First Clinical Proof • First clinical validation of the Recursion OS • Recursion’s phenotypic platform uncovered that selective MEK1/2 inhibition rescues cells from the loss of APC, revealing REC-4881 as a targeted therapeutic candidate for FAP • First to clinically investigate MEK1/2 inhibition for the treatment of FAP • Demonstrates Recursion’s ability to translate unbiased phenotypic and mechanistic insights into potentially differentiated therapeutics
REC-4881: Phase 1b/2 data review and next steps (I/II) 29 Rapid and Durable Reductions in Polyp Burden at 4 mg • Safety consistent with MEK inhibitors; low rate of Grade 3 events; no Grade 4/5 • Rapid and substantial clinical activity with 43% median reduction in 3 months; 75% of patients responded • Durable effect with 53% median reduction after 3 months off therapy; 82% of patients responded despite being off treatment • Responses seen in both Upper GI and Lower GI disease, with 40% Spigelman downstaging by Week 13 • Natural history shows patients see polyp burden progression when left untreated with 87% of patients experience significant annual polyp growth, 10% remained stable, and 3% showed modest reduction • Current investigational agents evaluated in separate studies generated approximately 17–29% reduction in polyp burden after 12 months of treatment. No off-treatment durability results were reported1 REC-4881 Potential First-in-Disease and Best-in-Class First Clinical Validation for Recursion OS Platform 1. Biodexa press release, June 24 2024

REC-4881: Phase 1b/2 data review and next steps (II/II) 30 Path Forward • Expanding population from ≥55 to ≥18 years old and further optimizing dosing schedule • Engage FDA (1H 2026) to define registration path informed by leveraging data generated thus far along with RWE assessments • Continue follow-up for evaluation of durability REC-4881 Potential First-in-Disease and Best-in-Class First Clinical Validation for Recursion OS Platform High Unmet Need in FAP • FAP affects ~50,000 patients in US/EU5 • Patients face life-long polyposis progression with near-100% CRC risk in the absence of surgery with no approved pharmacotherapies • Current sequential surgical treatments lead to substantial loss in quality of life and fail to target the underlying disease biology

31 Thank you to all the trial patients and their families and clinical trial sites

Fireside Chat Najat Khan, PhD | Chief R&D Officer and Commercial Officer, Incoming CEO Jessica Stout, DO | Asst Clinical Professor, University of Utah Al Cohen, MD, FACS, FASCRS | Former Chief of Colorectal Service, Memorial Sloan-Kettering Cancer Center David Mauro, MD, PhD | Chief Medical Officer
Fireside chat: Today’s speakers 33 Najat Khan Chief R&D Officer and Commercial Officer, Incoming CEO • Former Chief Data Science Officer & SVP, Global Head of R&D Strategy, Portfolio & Operations at Johnson & Johnson Innovative Medicine • Former Partner & Senior Principal at Boston Consulting Group, advising leading biopharma and healthcare organizations on strategy, operations, and innovation David Mauro, MD, PhD Chief Medical Officer Recursion • Former CMO Codiak BioSciences & Checkmate Pharmaceuticals • Former ED of Oncology Merck • Over 20 years of experience in oncology drug development, incl. spearheading early- and late-stage development for more than 25 INDs over the past decade Jessica Stout, DO Assistant Clinical Professor University of Utah School of Medicine • Double board-certified in Internal Medicine and Gastroenterology • Specializes in complex gastrointestinal disease management incl. long-term, multi-organ management of FAP Alfred M. Cohen, MD, FACS, FASCRS Former Chief of Colorectal Service Memorial Sloan-Kettering Cancer Center • Former CMO Cancer Prevention Pharmaceuticals • Former Director, CEO at Lucille P. Markey Cancer Center at University of Kentucky • >50 years of experience in CRC treatment & research, with clinical focus on QoL in patients with CRC, Lynch syndrome, and FAP

Event, n (%) REC-4881 Ph 1b and 2 4 mg (N=19 safety evaluable)1 Any TEAE related to study drug (TRAE) 18 (94.7) Grade 4/5 TRAE 0 Grade 3 TRAE 3 (15.8) Discontinuation due to TRAE 4 (21.1) Dose interruption due to TRAE 2 (10.5) Dose modification due to TRAE 0 REC-4881 Safety 4 mg dose: Safety profile consistent with MEK1/2 inhibition • Most common TRAEs Grade 1/2: • Dermatitis acneiform (57.9%; 52.6% Grade 1/2, 5.3% Grade 3) • Blood CPK increase (36.8%; 26.3% Grade 1/2, 10.5% Grade 3) • Rash (31.6%; all Grade 1/2) • Diarrhea (26.3%; all Grade 1/2) • LVEF decrease (10.5%; Grade 2)2 • Low rates of Grade 3 TRAEs, no Grade 4/5 events: • Grade 3 (n=3): Dermatitis acneiform (5.3%, n=1), blood CPK increase (10.5%, n=2) • Discontinuations (n=4) due to: • Grade 1 (n=1): 1 diarrhea • Grade 2 (n=3): 1 retinopathy, 1 rash, 1 hypertension • 92% common TRAEs are Grade 1/2 • 72% resolved within 12 weeks3 Data Cutoff Date: 2025-11-25 Note: For 8 mg (N=2, only 1 efficacy evaluable), N=1 (50%) exhibited Grade 2 TRAEs (rash), no Grade 3 TRAEs 1. For 4 mg, N=5 patients in Phase 1b and N=14 patients in Phase 2 were dosed; safety data cutoff date of 2025-11-25 2. Decrease was transient, patients were asymptomatic and 1 recovered following drug withdrawal. Based on using absolute percent change in LVEF, which is the widely used approach to measure changes in trials, for MEK inhibitors, etc. 3. Resolved: 96% Grade 1/2, 4% Grade 3; Unresolved: 71% Grade 1/2, 29% Grade 3 (e.g., CPK, Dermatitis Acneiform, rash)
Percent (%) change from baseline calculates the change between post-resection value from screening visit to the pre-resection value at Week 13/EOT visit. Subjects with absolute value of 0 at baseline are not displayed. Note: Polyp burden defined as the sum of all diameters of polyps in the GI 1. Following the March data cut, a quality review identified suboptimal bowel preparation at baseline. To ensure an accurate, like-for-like assessment, polyp burden was re-evaluated using video review restricted to the clean distal LGI segments matched to the same anatomical regions at Weeks 13 and 25 2. Patient reached W25 but did not perform W25 Assessment REC-4881 4mg dose level % change from baseline in total polyp burden at Week 13/EOT (12 weeks on therapy) % c h a n g e f ro m b a s e li n e 1 2 Data Cutoff Date: 2025-11-25
REC-4881 4mg dose level % change from baseline in total polyp burden at Week 25 (12 weeks off therapy) % c h a n g e f ro m b a s e li n e Percent (%) change from baseline calculates the change between post-resection value from screening visit to the pre-resection value at Week 25/EOT visit. Note: Polyp burden defined as the sum of all diameters of polyps in the GI 1. Non-responder with 233% increase – polyp burden increased from 3mm to 10mm due to one polyp growth at Week 25 1 Data Cutoff Date: 2025-11-25
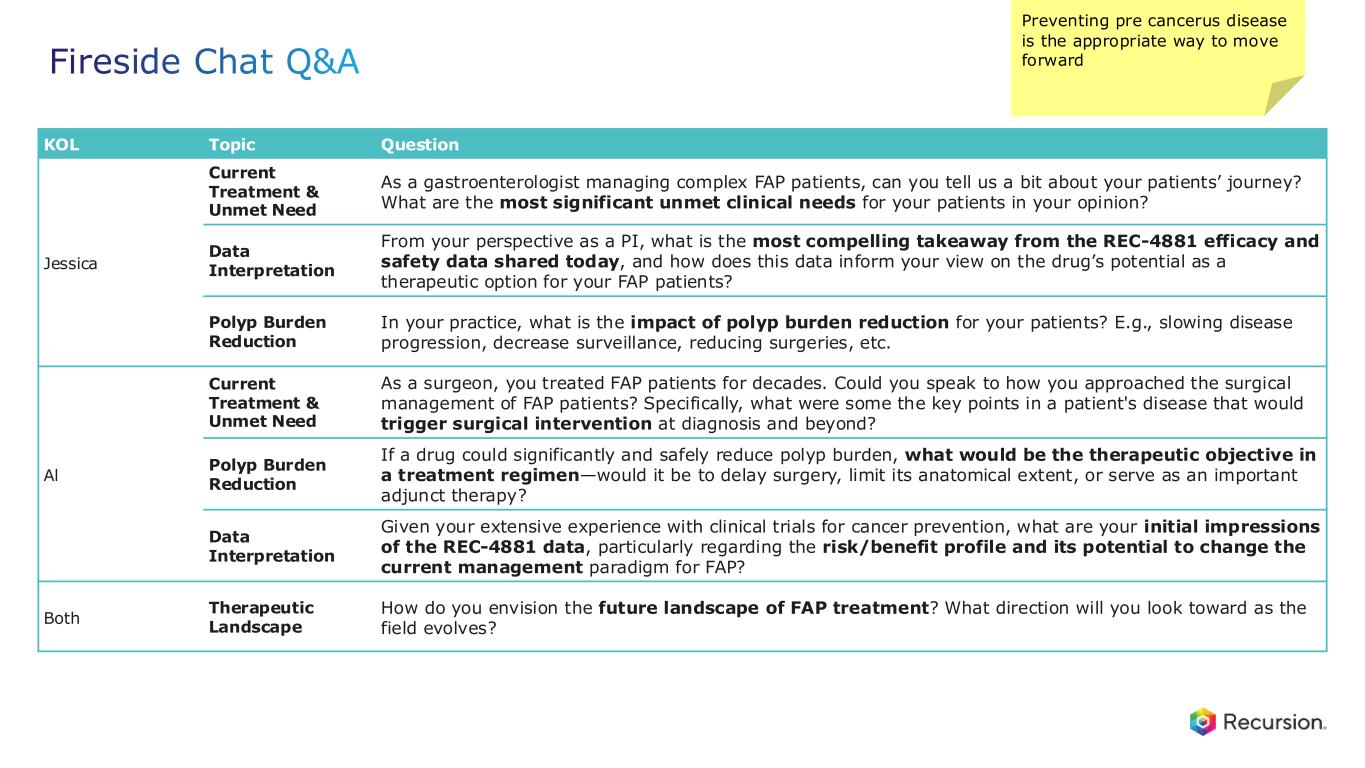
Fireside Chat Q&A KOL Topic Question Jessica Current Treatment & Unmet Need As a gastroenterologist managing complex FAP patients, can you tell us a bit about your patients’ journey? What are the most significant unmet clinical needs for your patients in your opinion? Data Interpretation From your perspective as a PI, what is the most compelling takeaway from the REC-4881 efficacy and safety data shared today, and how does this data inform your view on the drug’s potential as a therapeutic option for your FAP patients? Polyp Burden Reduction In your practice, what is the impact of polyp burden reduction for your patients? E.g., slowing disease progression, decrease surveillance, reducing surgeries, etc. Al Current Treatment & Unmet Need As a surgeon, you treated FAP patients for decades. Could you speak to how you approached the surgical management of FAP patients? Specifically, what were some the key points in a patient's disease that would trigger surgical intervention at diagnosis and beyond? Polyp Burden Reduction If a drug could significantly and safely reduce polyp burden, what would be the therapeutic objective in a treatment regimen—would it be to delay surgery, limit its anatomical extent, or serve as an important adjunct therapy? Data Interpretation Given your extensive experience with clinical trials for cancer prevention, what are your initial impressions of the REC-4881 data, particularly regarding the risk/benefit profile and its potential to change the current management paradigm for FAP? Both Therapeutic Landscape How do you envision the future landscape of FAP treatment? What direction will you look toward as the field evolves? NK 07-Dec-25: Surgical – major intervention (endoscopically and surgically) and don’t prophacalytically – patients with family history, surveillance Preventing pre cancerus disease is the appropriate way to move forward
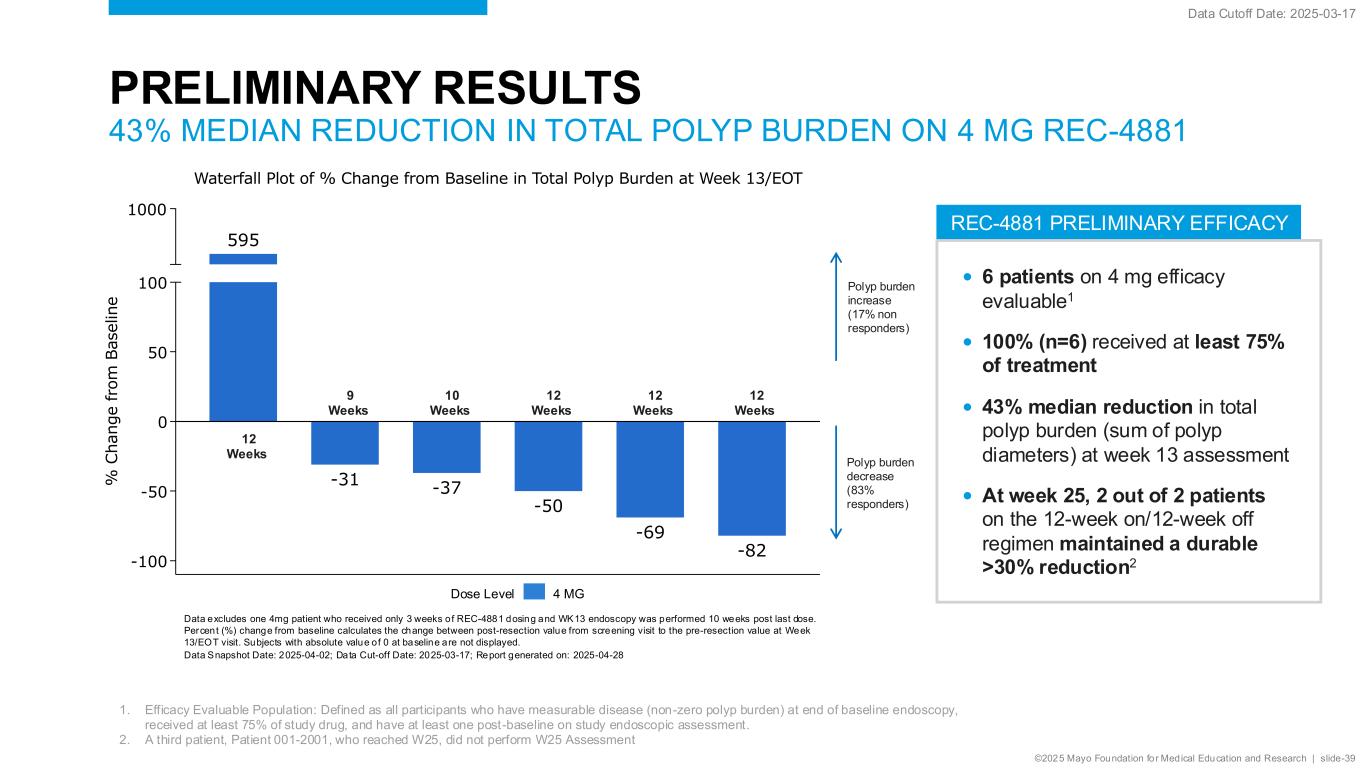
©2025 Mayo Foundation for Medical Education and Research | slide-39 REC-4881 PRELIMINARY EFFICACY PRELIMINARY RESULTS 43% MEDIAN REDUCTION IN TOTAL POLYP BURDEN ON 4 MG REC-4881 • 6 patients on 4 mg efficacy evaluable1 • 100% (n=6) received at least 75% of treatment • 43% median reduction in total polyp burden (sum of polyp diameters) at week 13 assessment • At week 25, 2 out of 2 patients on the 12-week on/12-week off regimen maintained a durable >30% reduction2 1. Efficacy Evaluable Population: Defined as all participants who have measurable disease (non-zero polyp burden) at end of baseline endoscopy, received at least 75% of study drug, and have at least one post-baseline on study endoscopic assessment. 2. A third patient, Patient 001-2001, who reached W25, did not perform W25 Assessment Data Cutoff Date: 2025-03-17 Polyp burden increase (17% non responders) Polyp burden decrease (83% responders) 12 Weeks 9 Weeks 10 Weeks 12 Weeks 12 Weeks 12 Weeks 595 -31 -37 -50 -69 -82 -100 -50 0 50 100 1000 % C h a n g e f ro m B a s e li n e Waterfall Plot of % Change from Baseline in Total Polyp Burden at Week 13/EOT Data excludes one 4mg patient who received only 3 weeks of REC-4881 dosing and WK13 endoscopy was performed 10 weeks post last dose. Percent (%) change from baseline calculates the change between post-resection value from screening visit to the pre-resection value at Week 13/EOT visit. Subjects with absolute value of 0 at baseline are not displayed. Data Snapshot Date: 2025-04-02; Data Cut-off Date: 2025-03-17; Report generated on: 2025-04-28 Dose Level 4 MG
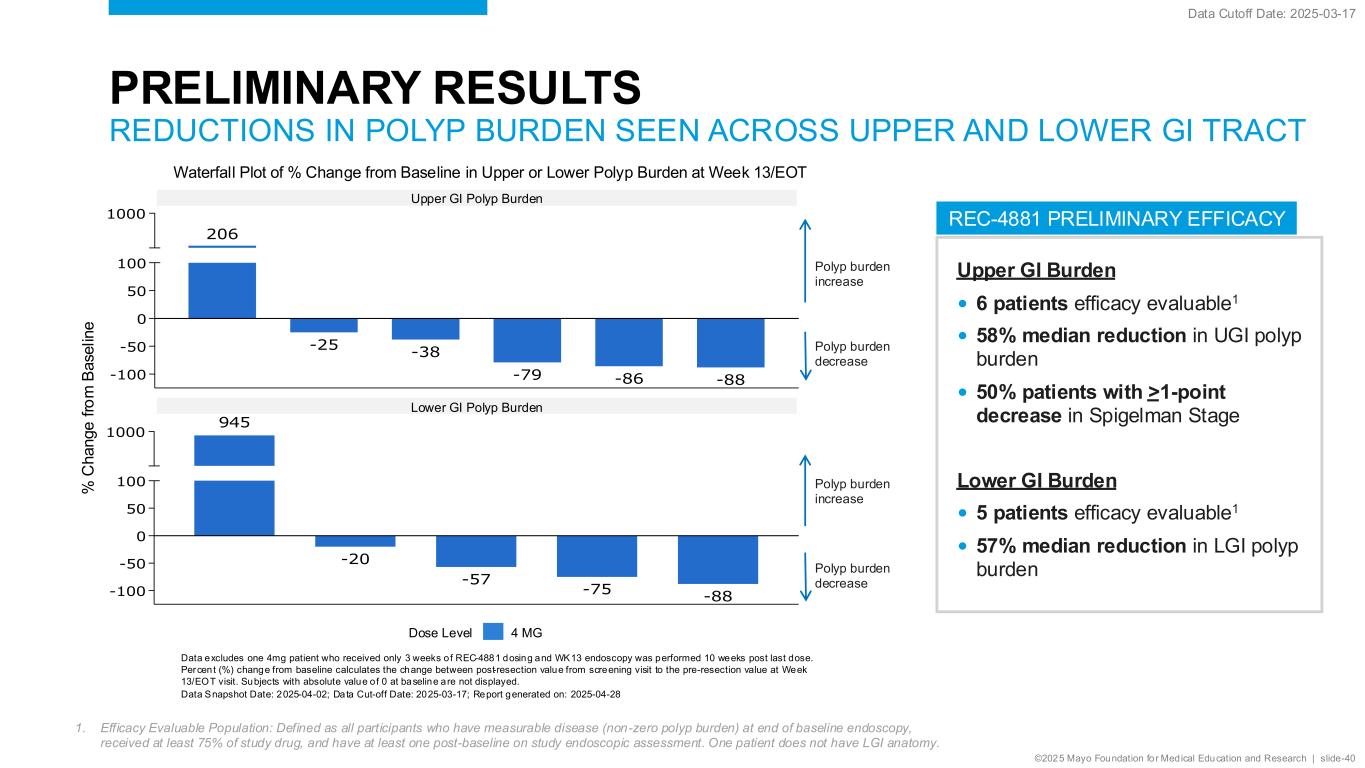
©2025 Mayo Foundation for Medical Education and Research | slide-40 REC-4881 PRELIMINARY EFFICACY PRELIMINARY RESULTS REDUCTIONS IN POLYP BURDEN SEEN ACROSS UPPER AND LOWER GI TRACT Upper GI Burden • 6 patients efficacy evaluable1 • 58% median reduction in UGI polyp burden • 50% patients with >1-point decrease in Spigelman Stage Lower GI Burden • 5 patients efficacy evaluable1 • 57% median reduction in LGI polyp burden Data Cutoff Date: 2025-03-17 1. Efficacy Evaluable Population: Defined as all participants who have measurable disease (non-zero polyp burden) at end of baseline endoscopy, received at least 75% of study drug, and have at least one post-baseline on study endoscopic assessment. One patient does not have LGI anatomy. -20 -57 -75 -88-100 -50 0 50 100 1000 % C h an g e fr o m B a se lin e 945 206 -25 -38 -79 -86 -88-100 -50 0 50 100 1000 % C h a n g e f ro m B a se lin e Lower GI Polyp Burden Upper GI Polyp Burden Waterfall Plot of % Change from Baseline in Upper or Lower Polyp Burden at Week 13/EOT Polyp burden increase Polyp burden decrease Polyp burden increase Polyp burden decrease % C h a n g e f ro m B a se lin e Data excludes one 4mg patient who received only 3 weeks of REC-4881 dosing and WK13 endoscopy was performed 10 weeks post last dose. Percent (%) change from baseline calculates the change between post-resection value from screening visit to the pre-resection value at Week 13/EOT visit. Subjects with absolute value o f 0 at baseline are not displayed. Data Snapshot Date: 2025-04-02; Data Cut-off Date: 2025-03-17; Report generated on: 2025-04-28 Dose Level 4 MG

©2025 Mayo Foundation for Medical Education and Research | slide-41 Proof of Concept of an AI based target selection and drug development in a rare disease with unmet need Preliminary REC-4881 safety • Generally well-tolerated in early analysis • TRAEs in 4 mg patients1 : rash (45.5%), dermatitis acneiform (45.5%), blood CPK increase (36.4%), diarrhea (27.3%) • Limited cardiac toxicity concern in Phase 2: 18% (N=2) patients reported G2 LVEF decrease Promising early efficacy results for 4 mg REC-4881 QD for 12 weeks showed: • 43% preliminary median total polyp burden reduction over 3 months • LGI polyp burden: 57% median reduction of • UGI polyp burden: 58% median reduction • 50% (N=3) reduced Spigelman stage by ≥1 point at Week 13 • Preliminary REC-4881 data suggests a high polyp burden reduction in a short timeframe (3 months). Other investigational agents have reported 20-30% reduction in 6 months2. Upcoming: • Continued enrollment across US centers with additional efficacy and safety analyses planned for H2, 2025 SUMMARY PRELIMINARY RESULTS AND NEXT STEPS 1. Phase 2 portion, N=11; Data cutoff 2025-03-17 2. Steinberg et al 2000 NEJM and Burke et al 2024 Gastroenterology
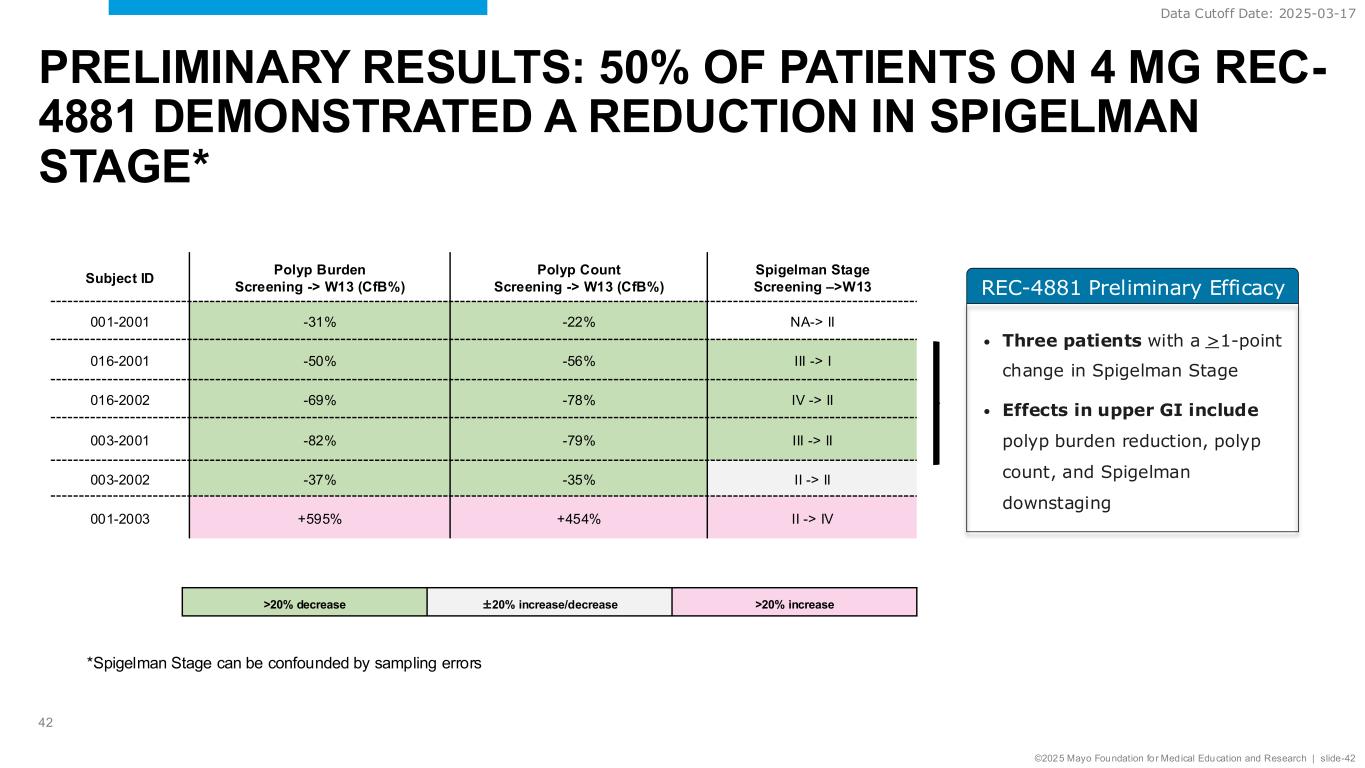
©2025 Mayo Foundation for Medical Education and Research | slide-42 REC-4881 Preliminary Efficacy PRELIMINARY RESULTS: 50% OF PATIENTS ON 4 MG REC- 4881 DEMONSTRATED A REDUCTION IN SPIGELMAN STAGE* 42 Subject ID Polyp Burden Screening -> W13 (CfB%) Polyp Count Screening -> W13 (CfB%) Spigelman Stage Screening –>W13 001-2001 -31% -22% NA-> II 016-2001 -50% -56% III -> I 016-2002 -69% -78% IV -> II 003-2001 -82% -79% III -> II 003-2002 -37% -35% II -> II 001-2003 +595% +454% II -> IV >20% decrease ±20% increase/decrease >20% increase • Three patients with a >1-point change in Spigelman Stage • Effects in upper GI include polyp burden reduction, polyp count, and Spigelman downstaging Data Cutoff Date: 2025-03-17 *Spigelman Stage can be confounded by sampling errors
